Limiting Reagent Lab Report

3 Limiting Reagent Lab Pdf Physical Chemistry Chemical Substances Chemistry lab report on limiting reagents experiment. includes procedure, data, calculations, and results. college level analytical chemistry. This laboratory report summarizes an experiment to determine the limiting reagent and percentage yield of a reaction between sodium carbonate and calcium nitrate.

Limiting Reagent Lab Report Mg Hcl Reaction Experiment summary: through the filtration of an aqueous solution containing sodium carbonate and barium acetate, one will effectively be able to determine the limiting reactant by testing which reactant the filtrate continues to react with in a test tube. In a separate reaction of calcium chloride and potassium hydroxide (performed in part b of this experiment), you will calculate the limiting reagent, theoretical yield, and percent yield after collection of the calcium hydroxide product. During a chemical reaction when two substances react, often times one reactant will be consumed before the other. the substance that is consumed first is called the limiting reactant. Limiting reactant (or limiting reagent) – the reactant that is completely used up during a chemical reaction, and therefore limits the amount of product that can be formed.

Lab 5 Limiting Reagent Analysis Chm 2045l Lab Report Studocu During a chemical reaction when two substances react, often times one reactant will be consumed before the other. the substance that is consumed first is called the limiting reactant. Limiting reactant (or limiting reagent) – the reactant that is completely used up during a chemical reaction, and therefore limits the amount of product that can be formed. Discuss how you identified the limiting reactant, how your experimental data compared with your theoretical predictions, and what might have caused any differences. To most clearly demonstrate how the extent of reaction in each of the experiments is determined by the limiting reactant, prepare two graphs of your experimental data. one graph should be for the reaction between hcl and naoh; the second graph should be for the reaction between h2so4 and naoh. If a reagent is present in a smaller quantity than the ratio requires, then it will be the limiting reagent. in our example, by calculating the moles of each reactant and dividing them by the stoichiometric ratio, we were able to find the limiting reagent. Conclusion: to conclude, this lab was the examination of the concept of limiting reactants using a fixed amount of one reagent and varying the amount of the second reagent for the chemical reaction.

Solved Stoichiometry And Limiting Reagent Lab Report Sheet Chegg Discuss how you identified the limiting reactant, how your experimental data compared with your theoretical predictions, and what might have caused any differences. To most clearly demonstrate how the extent of reaction in each of the experiments is determined by the limiting reactant, prepare two graphs of your experimental data. one graph should be for the reaction between hcl and naoh; the second graph should be for the reaction between h2so4 and naoh. If a reagent is present in a smaller quantity than the ratio requires, then it will be the limiting reagent. in our example, by calculating the moles of each reactant and dividing them by the stoichiometric ratio, we were able to find the limiting reagent. Conclusion: to conclude, this lab was the examination of the concept of limiting reactants using a fixed amount of one reagent and varying the amount of the second reagent for the chemical reaction.

Limiting Reactant Lab Report Pdf If a reagent is present in a smaller quantity than the ratio requires, then it will be the limiting reagent. in our example, by calculating the moles of each reactant and dividing them by the stoichiometric ratio, we were able to find the limiting reagent. Conclusion: to conclude, this lab was the examination of the concept of limiting reactants using a fixed amount of one reagent and varying the amount of the second reagent for the chemical reaction.
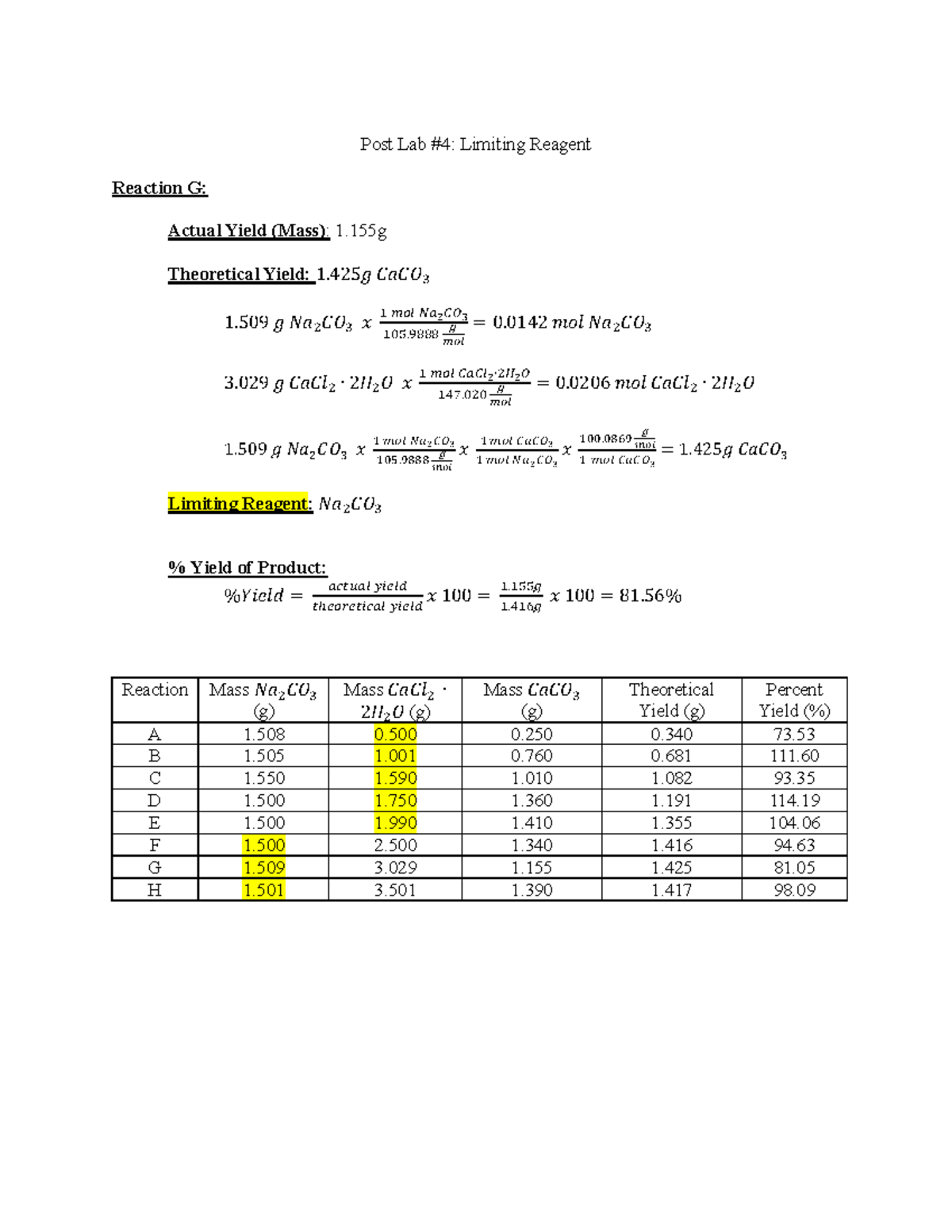
Lab Report 4 Post Lab Limiting Reagent Post Lab 4 Limiting
Comments are closed.