Lecture 10 Chem 102 E Aqueous Equilibria Youtube

Lecture 5 General Chemistry Ii Chem 102 Youtube Lecture 10: chem 102 e aqueous equilibria ahmad alakraa 3.42k subscribers subscribe. Numerade's aqueous equilibria: understanding the balance of solutions lectures chemistry 102 course focuses on the fundamental concepts of aqueous equilibria: understanding the balance of solutions.
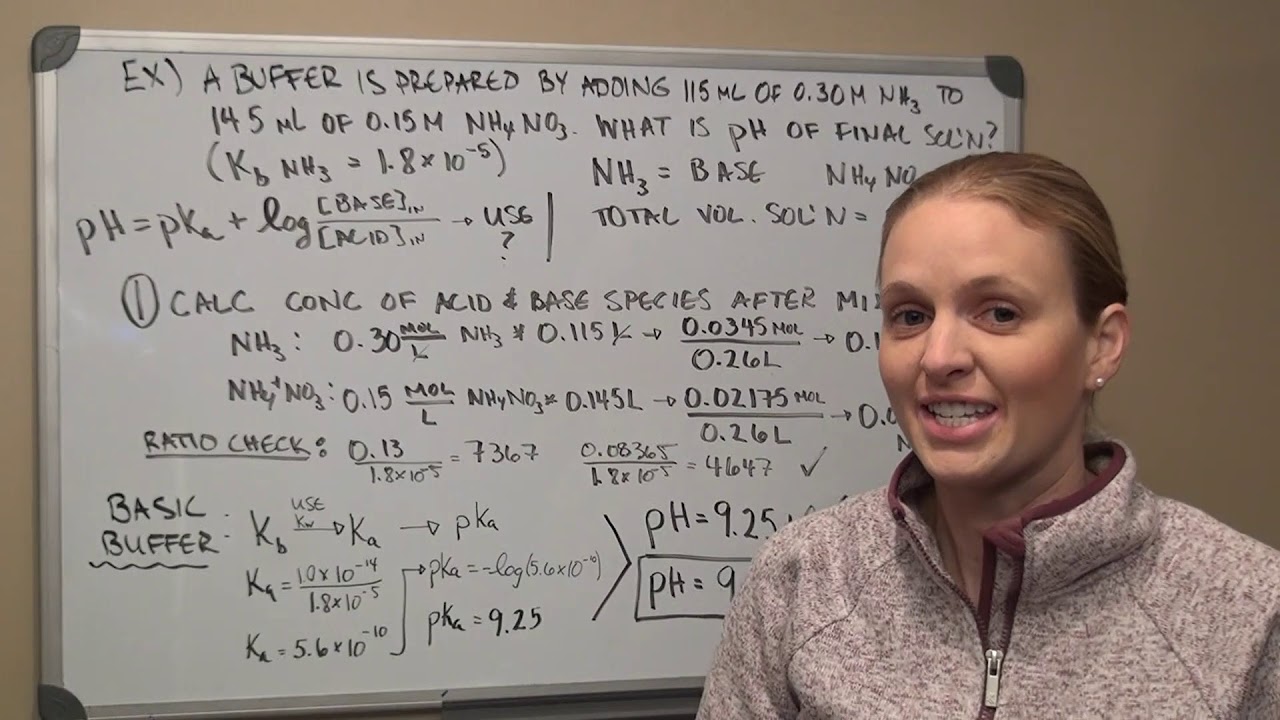
Aqueous Ionic Equilibria Lecture 2 Youtube Lecture 10 covers aqueous ionic equilibria, including buffer preparation, ph calculation using the henderson hasselbalch equation, and titration applications. it discusses solubility product constants (ksp), the common ion effect, and the impact of ph on solubility, as well as complex ion equilibria and their effects on solubility. Answers to math quiz this quiz illustrates the level of math is used in chem 102. Studying chem102 introductory university chemistry ii at university of alberta? on studocu you will find 150 lecture notes, practice materials, practical, mandatory. Calculate the concentration of cu 2 at equilibrium when nh 3 is added to a 0.010m cucl 2 solution to produce an equilibrium concentration of [nh 3] = 0.02m. neglect the volume change when the ammonia is added.

As Level Chemistry Equilibria Part 10 Youtube Studying chem102 introductory university chemistry ii at university of alberta? on studocu you will find 150 lecture notes, practice materials, practical, mandatory. Calculate the concentration of cu 2 at equilibrium when nh 3 is added to a 0.010m cucl 2 solution to produce an equilibrium concentration of [nh 3] = 0.02m. neglect the volume change when the ammonia is added. Description: uci chem 1c is the third and final quarter of general chemistry series and covers the following topics: equilibria, aqueous acid base equilibria, solubility equilibria, oxidation reduction reactions, electrochemistry; kinetics; special topics. The document explains the concept of equilibrium in chemistry, detailing physical and chemical equilibrium, the law of mass action, and how to calculate equilibrium constants (kc and kp). Subscribed 71 2k views 1 year ago chem 102 spring 2024 acids & bases; aqueous equilibria; ph calculations; salts acidity & basicity buffered solutions; buffering capacity more. The chemistry archive contains legacy chemistry content, and is not being updated with new content. for our most up to date, mastery enabled courses, check out middle school chemistry, high school chemistry and ap chemistry!.

A2 Chemical Equilibria Revision Class Lecture 1 Youtube Description: uci chem 1c is the third and final quarter of general chemistry series and covers the following topics: equilibria, aqueous acid base equilibria, solubility equilibria, oxidation reduction reactions, electrochemistry; kinetics; special topics. The document explains the concept of equilibrium in chemistry, detailing physical and chemical equilibrium, the law of mass action, and how to calculate equilibrium constants (kc and kp). Subscribed 71 2k views 1 year ago chem 102 spring 2024 acids & bases; aqueous equilibria; ph calculations; salts acidity & basicity buffered solutions; buffering capacity more. The chemistry archive contains legacy chemistry content, and is not being updated with new content. for our most up to date, mastery enabled courses, check out middle school chemistry, high school chemistry and ap chemistry!.
Chem 102 Introduction Youtube Subscribed 71 2k views 1 year ago chem 102 spring 2024 acids & bases; aqueous equilibria; ph calculations; salts acidity & basicity buffered solutions; buffering capacity more. The chemistry archive contains legacy chemistry content, and is not being updated with new content. for our most up to date, mastery enabled courses, check out middle school chemistry, high school chemistry and ap chemistry!.
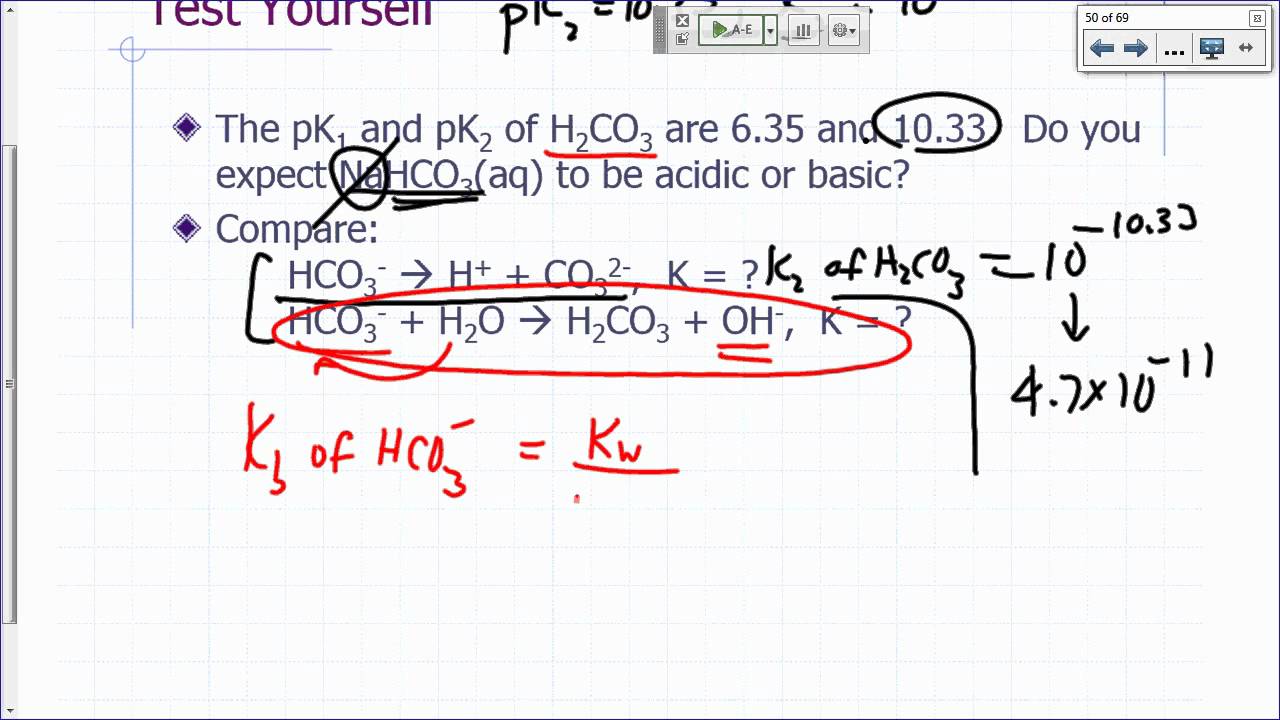
General Chemistry Lecture Aqueous Equilibria Part 3 Youtube
Comments are closed.