Aqueous Ionic Equilibria Lecture 2 Youtube

Ionic Equilibria Pdf Acid Aqueous Solution Henderson hasselbalch equation calculations of buffers, buffer effectiveness: capacity and range, acid base titrations, weak acid strong base titrations, s. Ionic equilibria of aqueous systems | lecture 2 | inorganic chemistry ii the statistics teacher 10.8k subscribers subscribed.

Ionic Equilibria Revision Notes Pdf In this video, we'll explore the fundamental concepts behind ionic equilibrium, including ph, acid base chemistry, buffer solutions, and titration curves. In this lecture, you will learn how to do calculations associated with strong acid strong base titrations at different points in the titration. … more. The equilibrium constant for a dissolution reaction, called the solubility product (ksp), is a measure of the solubility of a compound. whereas solubility is usually expressed in terms of mass of solute per 100 ml of solvent, ksp is defined in terms of the molar concentrations of the component ions. Lecture 37: brondsted lowry acid base equlibrium in every bronsted lowry equilibrium: each acid has a conjugate base each base has a conjugate acid. difference between acid & conjugate base is one h .

Ionic Equilibrium Lecture 14 Notes Pdf Pdf Buffer Solution Ph The equilibrium constant for a dissolution reaction, called the solubility product (ksp), is a measure of the solubility of a compound. whereas solubility is usually expressed in terms of mass of solute per 100 ml of solvent, ksp is defined in terms of the molar concentrations of the component ions. Lecture 37: brondsted lowry acid base equlibrium in every bronsted lowry equilibrium: each acid has a conjugate base each base has a conjugate acid. difference between acid & conjugate base is one h . The document provides an overview of electrolytes, acids, and bases, detailing their classifications as strong and weak electrolytes according to their ionization in aqueous solutions. Ionic equilibrium involves the balance between the concentrations of ions in a solution. this concept is essential in understanding how ions interact and maintain a stable environment in aqueous solutions. At equilibrium, an aqueous solution of weak acid contains a mixture of non ionized acid molecule and its conjugate base, respectively. On studocu you find all the lecture notes, summaries and study guides you need to pass your exams with better grades.

Ionic Equilibrium Youtube The document provides an overview of electrolytes, acids, and bases, detailing their classifications as strong and weak electrolytes according to their ionization in aqueous solutions. Ionic equilibrium involves the balance between the concentrations of ions in a solution. this concept is essential in understanding how ions interact and maintain a stable environment in aqueous solutions. At equilibrium, an aqueous solution of weak acid contains a mixture of non ionized acid molecule and its conjugate base, respectively. On studocu you find all the lecture notes, summaries and study guides you need to pass your exams with better grades.

Ionic Equilibria 1 A2 Level Chemistry Youtube At equilibrium, an aqueous solution of weak acid contains a mixture of non ionized acid molecule and its conjugate base, respectively. On studocu you find all the lecture notes, summaries and study guides you need to pass your exams with better grades.
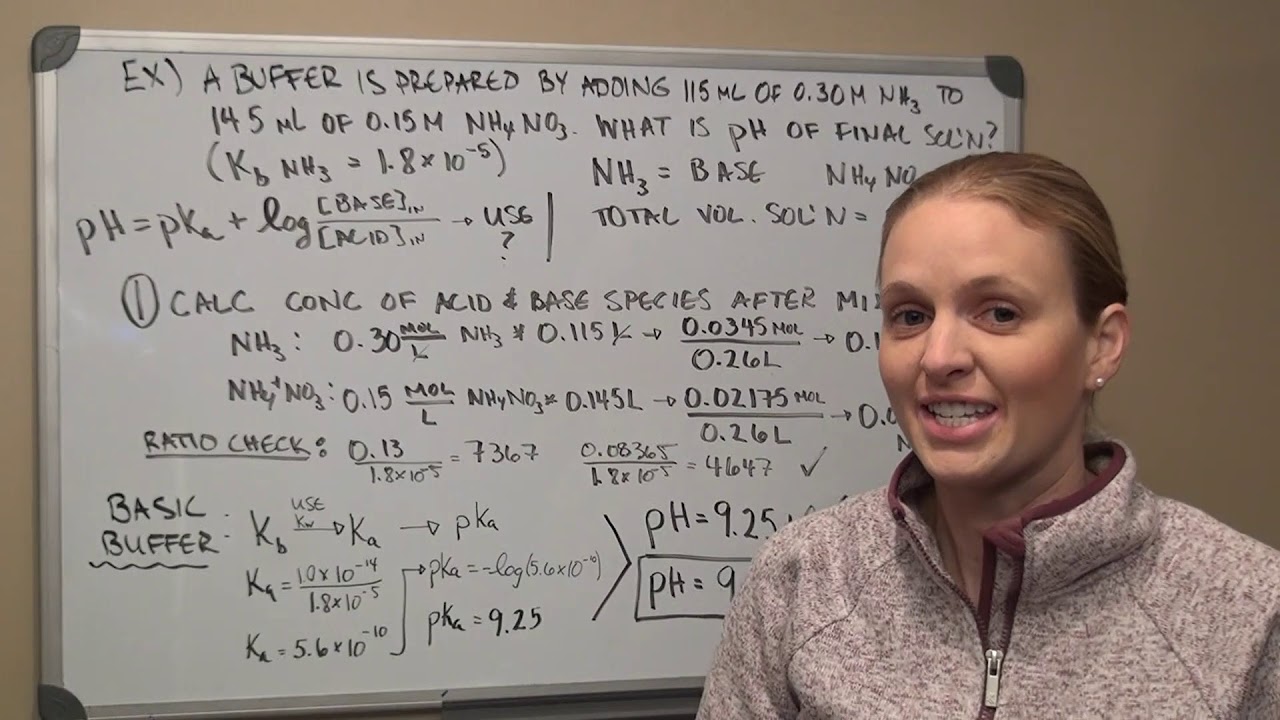
Aqueous Ionic Equilibria Lecture 2 Youtube
Comments are closed.