Lab 4 Limiting Reactant

Limiting Reactant Lab Download Free Pdf Sodium Bicarbonate Limiting reactant (or limiting reagent) – the reactant that is completely used up during a chemical reaction, and therefore limits the amount of product that can be formed. In a chemical reaction, the limiting reactant is the reactant that is completely used up, thus limiting the amount of product that can be produced, and the excess reactant is the reactant that is leftover.

Lab 4 Limiting Reactant Docx Lab 4 Limiting Reactant And Excess To determine which of the reactants was limiting, separate portions of the filtrate will be tested with the two reactants. the reactants that yields a positive result will have been the limiting reactant. Follow the instructions in the procedure to complete each part of the simulation. when instructed to record your observations, record data, or complete calculations, record them for your own records in order to use them later to complete the post lab assignment. This page details a laboratory experiment using sodium bicarbonate and vinegar in balloons and bottles labeled a, b, and c. students will measure baking soda and vinegar, calculate moles, and perform the reaction. During a chemical reaction when two substances react, often times one reactant will be consumed before the other. the substance that is consumed first is called the limiting reactant.
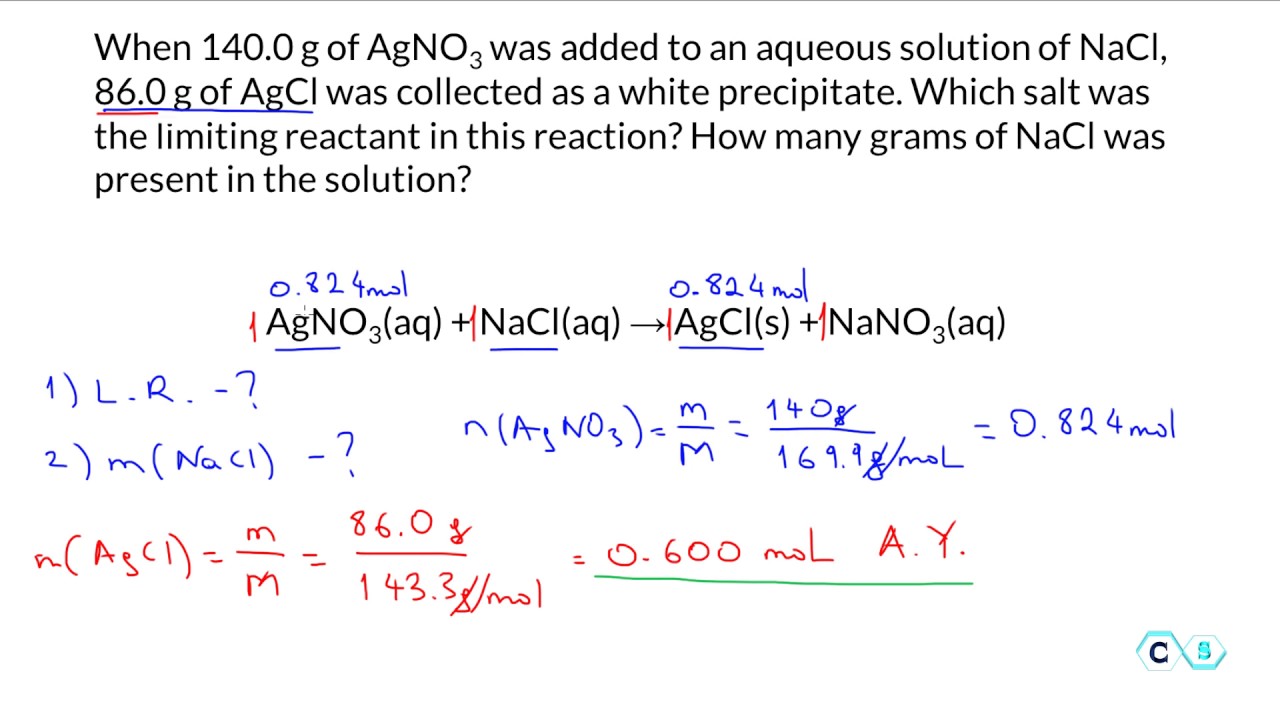
Limiting Reactant Lab Activity At Brooke Rentoul Blog This page details a laboratory experiment using sodium bicarbonate and vinegar in balloons and bottles labeled a, b, and c. students will measure baking soda and vinegar, calculate moles, and perform the reaction. During a chemical reaction when two substances react, often times one reactant will be consumed before the other. the substance that is consumed first is called the limiting reactant. The concept of limiting reactant is very important in the study of the stoichiometry of chemical reactions. the limiting reactant is the reactant that controls the amount of product possible for a process because once the limiting reactant has been consumed, no further reaction can occur. Limiting reactant experiment 4 a limiting reactant is the reagent that is completely consumed during a chemical reaction. once this reagent is consumed the reaction stops. an excess reagent is the reactant that is left over once the limiting reagent is consumed. In this activity, students will practice drawing particulate diagrams to help them determine the limiting reactant given a certain number of reactant molecules. When one reactant is used up, no more products can form. if one reactant gets used up before another reactant in a chemical reaction, we call the reactant, which is used up the limiting reactant. the reactant that is left over is called the excess reactant.

Limiting Reactant Explained Step By Step Guide Practice Problems The concept of limiting reactant is very important in the study of the stoichiometry of chemical reactions. the limiting reactant is the reactant that controls the amount of product possible for a process because once the limiting reactant has been consumed, no further reaction can occur. Limiting reactant experiment 4 a limiting reactant is the reagent that is completely consumed during a chemical reaction. once this reagent is consumed the reaction stops. an excess reagent is the reactant that is left over once the limiting reagent is consumed. In this activity, students will practice drawing particulate diagrams to help them determine the limiting reactant given a certain number of reactant molecules. When one reactant is used up, no more products can form. if one reactant gets used up before another reactant in a chemical reaction, we call the reactant, which is used up the limiting reactant. the reactant that is left over is called the excess reactant.

Limiting Reactant Lab Chemistry Experiment Percent Yield In this activity, students will practice drawing particulate diagrams to help them determine the limiting reactant given a certain number of reactant molecules. When one reactant is used up, no more products can form. if one reactant gets used up before another reactant in a chemical reaction, we call the reactant, which is used up the limiting reactant. the reactant that is left over is called the excess reactant.
Comments are closed.