Joule Thomson Effect Definition Example Coefficient Derivation

Joule Thomson Effect Pdf Gases Mechanical Engineering This equation can be used to obtain joule–thomson coefficients from the more easily measured isothermal joule–thomson coefficient. it is used in the following to obtain a mathematical expression for the joule–thomson coefficient in terms of the volumetric properties of a fluid. The joule thomson coefficient can be derived using thermodynamic relationships and is defined as the change in temperature during an isenthalpic process (constant enthalpy) due to a pressure drop:.

Joule Thomson Effect Coefficient Calculation For Co2 And N2 It is also known as joule kelvin or kelvin joule effect. their theory states that changes in the pressure of the valve can lead to temperature fluctuations. read on to learn more about its concept along with derivation and applications. In thermodynamics, the effect called joule thomson effect was discovered in 1852. this effect was named after two physicists james prescott joule (1818 1889) and william thomson (1824 1907). Some gases, such as hydrogen and helium, will experience a warming effect upon expansion under conditions near room temperature and pressure. the direction of temperature change can be determined by measuring the joule thomson coefficient, μ j t. this coefficient has the definition μ j t ≡ (∂ t ∂ p) h. What is the joule thomson coefficient (μ jt) and its formula? the joule thomson coefficient (μjt) measures the rate of temperature change of a gas with respect to pressure at constant enthalpy. • positive μ jt means cooling; negative means heating during expansion.
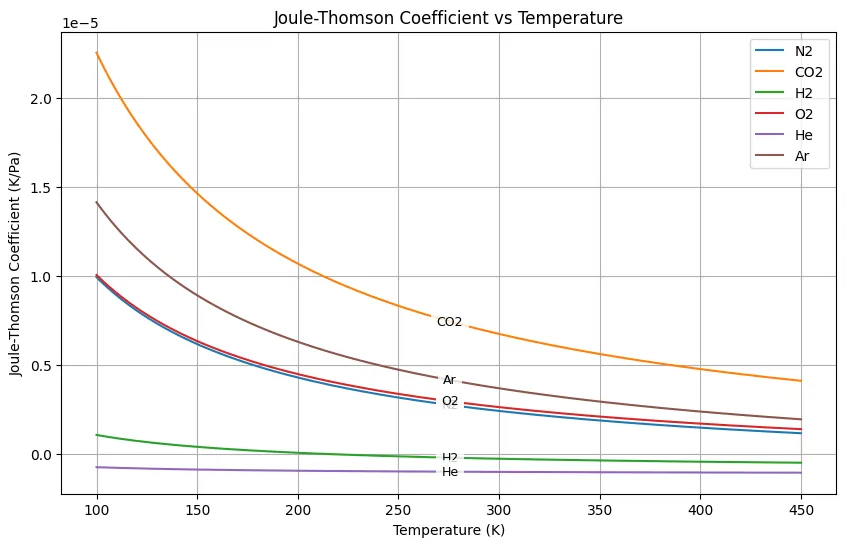
Joule Thomson Effect Coefficient Calculation For Co2 And N2 Some gases, such as hydrogen and helium, will experience a warming effect upon expansion under conditions near room temperature and pressure. the direction of temperature change can be determined by measuring the joule thomson coefficient, μ j t. this coefficient has the definition μ j t ≡ (∂ t ∂ p) h. What is the joule thomson coefficient (μ jt) and its formula? the joule thomson coefficient (μjt) measures the rate of temperature change of a gas with respect to pressure at constant enthalpy. • positive μ jt means cooling; negative means heating during expansion. What is joule thomson effect? the joule thomson effect also known as kelvin–joule effect or joule kelvin effect is the change in fluid’s temperature as it flows from a higher pressure region to lower pressure. This python code helps user to plot the joule–thomson coefficient 𝜇jt as a function of temperature for various real gases, like nitrogen, carbon dioxide, hydrogen, oxygen, helium, and argon. Equation (5) is the general thermodynamic expression for the joule –thomson co efficient of the gas. thus for an ideal gas, joule –thomson co efficient is zero, i.e., an ideal gas does not exhibit the joule thomson effect. the term a pv & ab pv2, we are dividing a small quantity by a large quantity. To assess whether a van der waals gas can be liquefied using the joule–thomson effect, it is necessary to evaluate the joule–thomson coefficient, μ. this is related to (∂v ∂t) p, which can be evaluated in the following way.
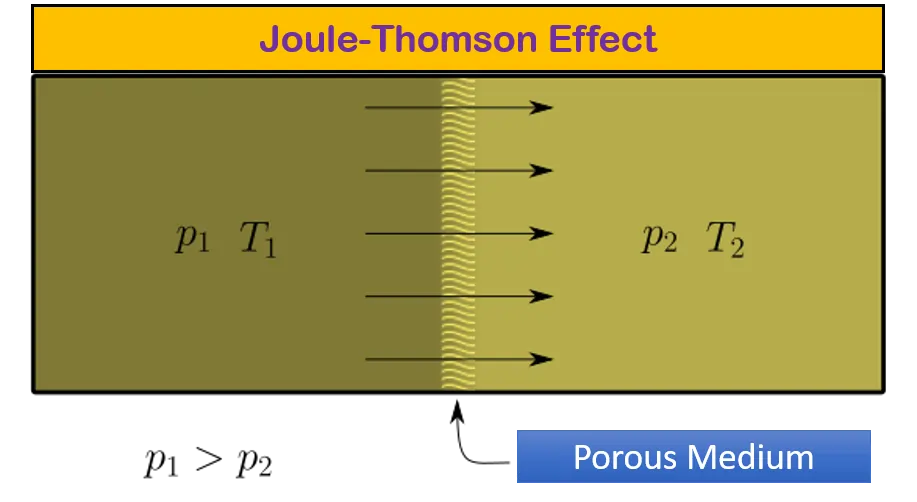
Joule Thomson Effect Coefficient Calculation For Co2 And N2 What is joule thomson effect? the joule thomson effect also known as kelvin–joule effect or joule kelvin effect is the change in fluid’s temperature as it flows from a higher pressure region to lower pressure. This python code helps user to plot the joule–thomson coefficient 𝜇jt as a function of temperature for various real gases, like nitrogen, carbon dioxide, hydrogen, oxygen, helium, and argon. Equation (5) is the general thermodynamic expression for the joule –thomson co efficient of the gas. thus for an ideal gas, joule –thomson co efficient is zero, i.e., an ideal gas does not exhibit the joule thomson effect. the term a pv & ab pv2, we are dividing a small quantity by a large quantity. To assess whether a van der waals gas can be liquefied using the joule–thomson effect, it is necessary to evaluate the joule–thomson coefficient, μ. this is related to (∂v ∂t) p, which can be evaluated in the following way.

Joule Thomson Effect Definition Joule Thomson Coefficient Equation (5) is the general thermodynamic expression for the joule –thomson co efficient of the gas. thus for an ideal gas, joule –thomson co efficient is zero, i.e., an ideal gas does not exhibit the joule thomson effect. the term a pv & ab pv2, we are dividing a small quantity by a large quantity. To assess whether a van der waals gas can be liquefied using the joule–thomson effect, it is necessary to evaluate the joule–thomson coefficient, μ. this is related to (∂v ∂t) p, which can be evaluated in the following way.

Joule Thomson Effect Definition Joule Thomson Coefficient
Comments are closed.