Intro Joule Thomson Coefficient

Lecture 25 Tchm The Joule Thomson Coefficient Pdf Physical A useful derivative for understanding cooling or heating of gases when they expand is the joule thomson coefficient (\ (\mu {jt}\)). this derivative describes how the temperature of a gas changes when …. The joule–thomson coefficient is defined as the temperature change of a nonideal gas when it expands from high pressure to low pressure under adiabatic conditions, represented mathematically as η = (∂t ∂p)ₕ.
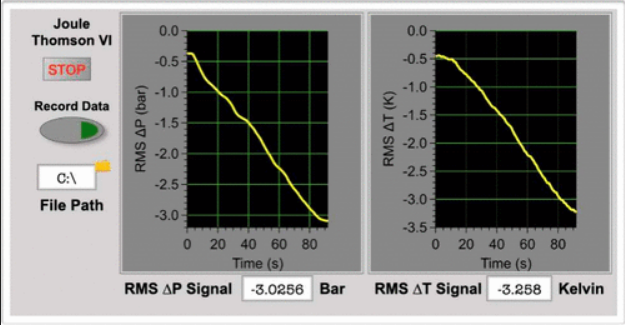
Measurement Of The Joule Thomson Coefficient Of Nitrogen And Carbon The rate of change of temperature with respect to pressure in a joule–thomson process (that is, at constant enthalpy ) is the joule–thomson (kelvin) coefficient . In thermodynamics, the effect called joule thomson effect was discovered in 1852. this effect was named after two physicists james prescott joule (1818 1889) and william thomson (1824 1907). The slope of a constant enthalpy is known as joule thomson coefficient, it is denoted by μ. for real gas, μ may be either positive or negative depending upon the thermodynamic state of the gas. The joule thomson coefficient is a crucial parameter in understanding the behavior of gases under various conditions. it is a measure of the change in temperature of a gas as it expands through a valve or a porous plug from a high pressure to a low pressure.
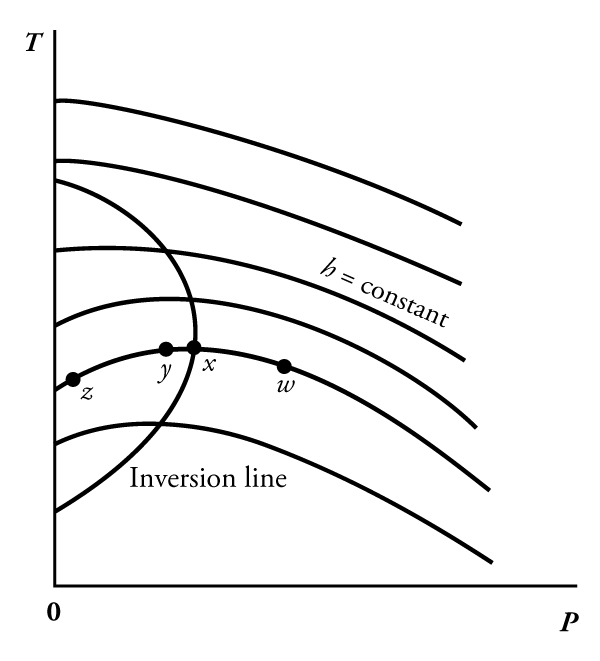
Thermodynamics Joule Thomson Coefficient Physics Stack Exchange The slope of a constant enthalpy is known as joule thomson coefficient, it is denoted by μ. for real gas, μ may be either positive or negative depending upon the thermodynamic state of the gas. The joule thomson coefficient is a crucial parameter in understanding the behavior of gases under various conditions. it is a measure of the change in temperature of a gas as it expands through a valve or a porous plug from a high pressure to a low pressure. The joule thomson experiment must be carefully distinguished from the adiabatic expansion of a gas in a piston and cylinder. the latter process normally leads to a cooling, on account of the decrease in internal energy consequent on the performance of work. What is the joule thomson coefficient (μ jt) and its formula? the joule thomson coefficient (μjt) measures the rate of temperature change of a gas with respect to pressure at constant enthalpy. • positive μ jt means cooling; negative means heating during expansion. Explore the joule thomson coefficient, the key to refrigeration. learn why real gases cool or heat upon expansion and its vast applications from cryogenics to cosmology. The joule thomson effect describes the change in temperature of a real gas or liquid when it is forced through a valve or porous plug while kept insulated so that no heat is exchanged with the environment.
Comments are closed.