Joule S Law Pdf

Joule S Law Pdf Electrical Conductor Heat James prescott joule (1818 1889), born into a well to do family prominent in the brewery industry, studied at manchester under dalton. at age twenty one he published the "i squared r" law which bears his name. Joule's law free download as word doc (.doc .docx), pdf file (.pdf), text file (.txt) or read online for free. joule's law describes the conversion of electrical energy into heat when an electric current flows through a conductor, due to collisions between electrons and atoms.

Joule S Law Of Heating Vector Illustration Stock Vector Adobe Stock Lesson 2: joule’s law and electric power a. electrical power • that as charges flow through a circuit device (like a resistor or lightbulb), they lose energy due to collisions with the atoms in the wire. The 1st law states ∆e = q w (1) is the same for all transformations leading form a given initial state to a final state (joule’s law), where e is the total energy (or internal energy, or just energy) of the td system. clearly, e, q and w are all measured in energy unit (si: joule). In this experiment heat is produced by passing a current through a resistor which is immeresed in a container of water. the purpose of this experiment is to compare the energy dissipated by the resistor to that absorbed by the water, thereby testing the principle of conservation of energy. the experimental arrangement is shown in figure 1. According to joule’s law, under these conditions the tem perature of the gas does not change, which implies that the kinetic energy of the molecules remains constant.
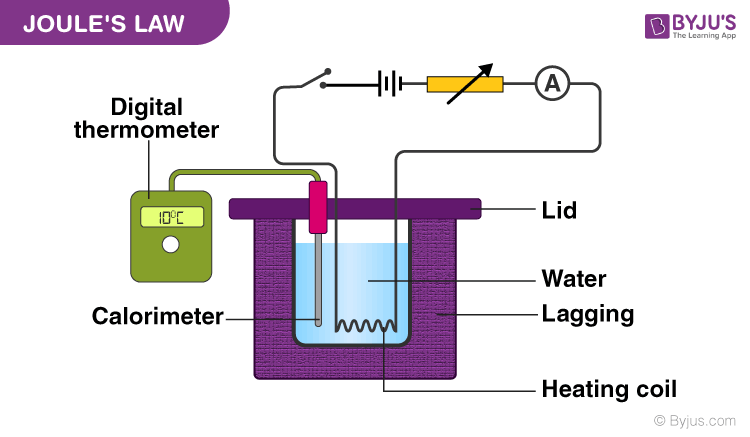
Joule S Law Of Heating And Joule S First Law Physics Byju S In this experiment heat is produced by passing a current through a resistor which is immeresed in a container of water. the purpose of this experiment is to compare the energy dissipated by the resistor to that absorbed by the water, thereby testing the principle of conservation of energy. the experimental arrangement is shown in figure 1. According to joule’s law, under these conditions the tem perature of the gas does not change, which implies that the kinetic energy of the molecules remains constant. Early distribution systems used a voltage of 2200 volts, increased to 2400 volts. as cities grew, most 2400 volt upgraded to 2400 4160 y three phase systems. most cities now have been converted higher voltage systems. Download as a pdf or view online for free. (1) first law : it states that the mass of substance deposited at the cathode during electrolysis is directly proportional to the quantity of electricity (total charge) passed through the electrolyte. This module explores two important mathematical relationships for electric circuits: ohm’s law and joule’s law. the first of these relates voltage and current to resistance, while the second relates those quantities to power.

Hukum Joule Pdf Early distribution systems used a voltage of 2200 volts, increased to 2400 volts. as cities grew, most 2400 volt upgraded to 2400 4160 y three phase systems. most cities now have been converted higher voltage systems. Download as a pdf or view online for free. (1) first law : it states that the mass of substance deposited at the cathode during electrolysis is directly proportional to the quantity of electricity (total charge) passed through the electrolyte. This module explores two important mathematical relationships for electric circuits: ohm’s law and joule’s law. the first of these relates voltage and current to resistance, while the second relates those quantities to power.

Joule S Law Pdf (1) first law : it states that the mass of substance deposited at the cathode during electrolysis is directly proportional to the quantity of electricity (total charge) passed through the electrolyte. This module explores two important mathematical relationships for electric circuits: ohm’s law and joule’s law. the first of these relates voltage and current to resistance, while the second relates those quantities to power.
Comments are closed.