Iq Oq Pq Validation Guideline Documents Pdf Verification And

Iq Oq Pq Validation Guideline Documents Pdf Verification And This guide outlines the protocols of installation qualification (iq), operational qualification (oq), and performance qualification (pq) essential for ensuring equipment in fda regulated industries meets quality assurance standards. Explore comprehensive pdf examples for iq, oq, and pq validation. download free templates and learn best practices for your validation process.

Iq Oq Pq Get Equipment Validation Right The First Time Scilife Unlock the secrets of quality assurance with our comprehensive guide to iq oq pq protocols. download our easy to follow pdf examples and templates to streamline your validation process, ensuring your projects meet industry standards with confidence!. Access validated iq oq pq templates and examples in pdf format from regulatory websites, industry forums, or professional associations like ispe or pda for practical guidance. Validation: iq, oq, pq iq, oq, pq are requirement based methods that facilitate the validation of a process. nitoring or measuring. these types of validations are performed to reduce production costs and ensure regulator. The validation activities and results, including the date and signature of the individual(s) approving the validation and where appropriate the major equipment validated, shall be documented.
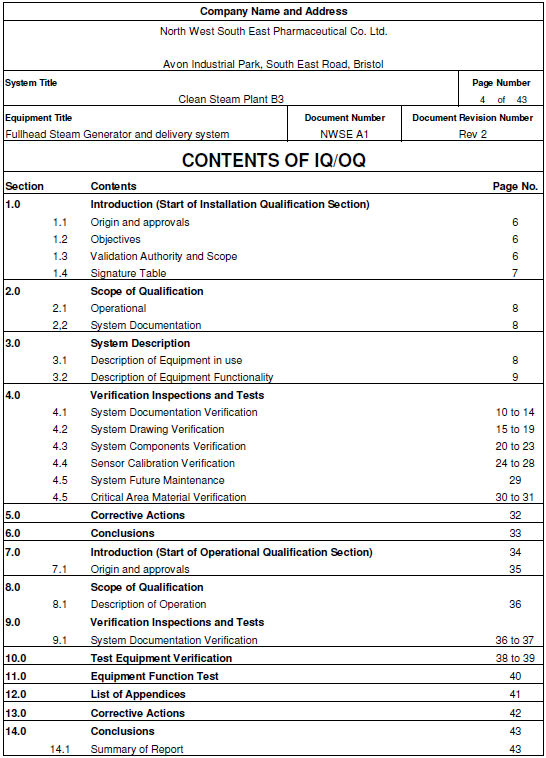
Iq Oq Pq Steam Quality Qualification Documentation Validation: iq, oq, pq iq, oq, pq are requirement based methods that facilitate the validation of a process. nitoring or measuring. these types of validations are performed to reduce production costs and ensure regulator. The validation activities and results, including the date and signature of the individual(s) approving the validation and where appropriate the major equipment validated, shall be documented. The objective of this protocol is to define the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the [insert system name and plant number] which will be located in the [insert area, packaging or manufacturing] at site [insert site name]. Our equipment qualification and validation service supports a broad range of equipment types. our suite of validation option packages will ensure that you comply with applicable installation, operational and performance qualification standards (iq oq pq). The iq outlines procedures used to confirm the instrument and requisite software are installed correctly, while the oq includes tests to check both the instrument and software are functioning as expected. We cover the regulatory background and industry standards that mandate iq oq pq, detailed descriptions of each qualification phase, and the challenges of manual processes.

Fda Part 11 Your Guide To Computer System Validation Csv Compliance The objective of this protocol is to define the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the [insert system name and plant number] which will be located in the [insert area, packaging or manufacturing] at site [insert site name]. Our equipment qualification and validation service supports a broad range of equipment types. our suite of validation option packages will ensure that you comply with applicable installation, operational and performance qualification standards (iq oq pq). The iq outlines procedures used to confirm the instrument and requisite software are installed correctly, while the oq includes tests to check both the instrument and software are functioning as expected. We cover the regulatory background and industry standards that mandate iq oq pq, detailed descriptions of each qualification phase, and the challenges of manual processes.
Comments are closed.