Iq Oq Pq A Quick Guide To Process Validation 47 Off

Iq Oq Pq A Quick Guide To Process Validation Pdf Verification Learn about process validation and its three phases: installation qualification (iq), operation qualification (oq), and performance qualification (pq). Iq, oq, pq: a quick guide to process validation free download as pdf file (.pdf), text file (.txt) or read online for free.

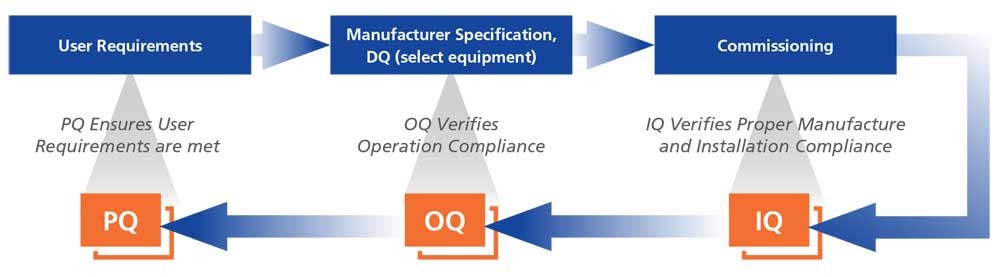
Iq Oq Pq Sop Pdf Verification And Validation Business Process This guide covers equipment qualification requirements, procedures, and best practices to meet fda process validation standards. pharmaceutical companies rely on iq, oq, pq to verify machine performance. Master iq, oq, and pq with this practical guide to equipment validation. learn the differences, see real world examples, and apply proven best practices. The concepts of installation qualification (iq), operational qualification (oq), and performance qualification (pq) are closely interlinked, forming the backbone of the equipment validation process. What is the primary difference between iq, oq, and pq in pharmaceutical labs? iq verifies correct installation, oq confirms the equipment operates within specified ranges, and pq demonstrates consistent performance for its intended routine use over time.

Guide To Iq Oq Pq For The Pharmaceutical Industry Pdf The concepts of installation qualification (iq), operational qualification (oq), and performance qualification (pq) are closely interlinked, forming the backbone of the equipment validation process. What is the primary difference between iq, oq, and pq in pharmaceutical labs? iq verifies correct installation, oq confirms the equipment operates within specified ranges, and pq demonstrates consistent performance for its intended routine use over time. The purpose of this sop is to outline the procedures for performing installation qualification (iq), operational qualification (oq), and performance qualification (pq) in manufacturing process validation. One of the key sets of protocols within equipment validation is installation qualification (iq), operational qualification (oq), and performance qualification (pq). This guide shows how to plan and execute iq, oq, and pq for pharmaceutical and pharmaceutical logistics environments – and how to keep equipment qualified with less rework and fewer interruptions. Get a basic guide to iq, oq, pq for the pharmaceutical, medical device, and diagnostic industries, including fda criteria and a model for resourcing.

Iq Oq Pq A Quick Guide To Process Validation 47 Off The purpose of this sop is to outline the procedures for performing installation qualification (iq), operational qualification (oq), and performance qualification (pq) in manufacturing process validation. One of the key sets of protocols within equipment validation is installation qualification (iq), operational qualification (oq), and performance qualification (pq). This guide shows how to plan and execute iq, oq, and pq for pharmaceutical and pharmaceutical logistics environments – and how to keep equipment qualified with less rework and fewer interruptions. Get a basic guide to iq, oq, pq for the pharmaceutical, medical device, and diagnostic industries, including fda criteria and a model for resourcing.
Iq Oq Pq A Quick Guide To Process Validation 40 Off This guide shows how to plan and execute iq, oq, and pq for pharmaceutical and pharmaceutical logistics environments – and how to keep equipment qualified with less rework and fewer interruptions. Get a basic guide to iq, oq, pq for the pharmaceutical, medical device, and diagnostic industries, including fda criteria and a model for resourcing.

Iq Oq Pq A Quick Guide To Process Validation 56 Off
Comments are closed.