Introduction To Limiting Reactant And Excess Reactant

Limiting Reactant And Excess Reactant Pptx Chemistry Science We learned that the limiting reactant is the reactant that limits the amount of product that can be made, while an excess reactant is one that is not entirely consumed. Limiting and excess reactants are key concepts in chemical reactions. they determine how much product can be made and which reactants will be left over. understanding these ideas is crucial for predicting reaction outcomes and optimizing processes.

Limiting Reactant And Excess Reactant Pptx Chemistry Science We will learn about limiting reactant and limiting reagent by comparing chemical reactions to cooking recipes and we will look at an actual stoichiometry problem. Identifying the limiting and excess reactants for a given situation requires computing the molar amounts of each reactant provided and comparing them to the stoichiometric amounts represented in the balanced chemical equation. This document discusses limiting and excess reactants in chemical reactions. it defines a limiting reactant as the reactant that is consumed first or ends the reaction. If these reactants are provided in any other amounts, one of the reactants will nearly always be entirely consumed, thus limiting the amount of product that may be generated. this substance is the limiting reactant, and the other substance is the excess reactant.

Limiting Reactant And Excess Reactant Pptx Chemistry Science This document discusses limiting and excess reactants in chemical reactions. it defines a limiting reactant as the reactant that is consumed first or ends the reaction. If these reactants are provided in any other amounts, one of the reactants will nearly always be entirely consumed, thus limiting the amount of product that may be generated. this substance is the limiting reactant, and the other substance is the excess reactant. Learn how to solve limiting reactant problems in stoichiometry by identifying which reactant limits the reaction and directly determines the product amount. this lesson offers clear methods, worked out examples, and guidance for confident problem solving. In a chemical reaction, the limiting reactant is the reactant that is completely used up, thus limiting the amount of product that can be produced, and the excess reactant is the reactant that is leftover. Learn how to identify limiting and excess reactants with our step by step stoichiometry guide. calculate theoretical yield easily. master your chemistry exams now!. Limiting reactant is also what prevents a reaction from continuing because there is none left. the limiting reactant may also be referred to as limiting reagent or limiting agent. the reactant that is not used up is referred to as the excess reactant.

Limiting Reactant And Excess Reactant Pptx Learn how to solve limiting reactant problems in stoichiometry by identifying which reactant limits the reaction and directly determines the product amount. this lesson offers clear methods, worked out examples, and guidance for confident problem solving. In a chemical reaction, the limiting reactant is the reactant that is completely used up, thus limiting the amount of product that can be produced, and the excess reactant is the reactant that is leftover. Learn how to identify limiting and excess reactants with our step by step stoichiometry guide. calculate theoretical yield easily. master your chemistry exams now!. Limiting reactant is also what prevents a reaction from continuing because there is none left. the limiting reactant may also be referred to as limiting reagent or limiting agent. the reactant that is not used up is referred to as the excess reactant.
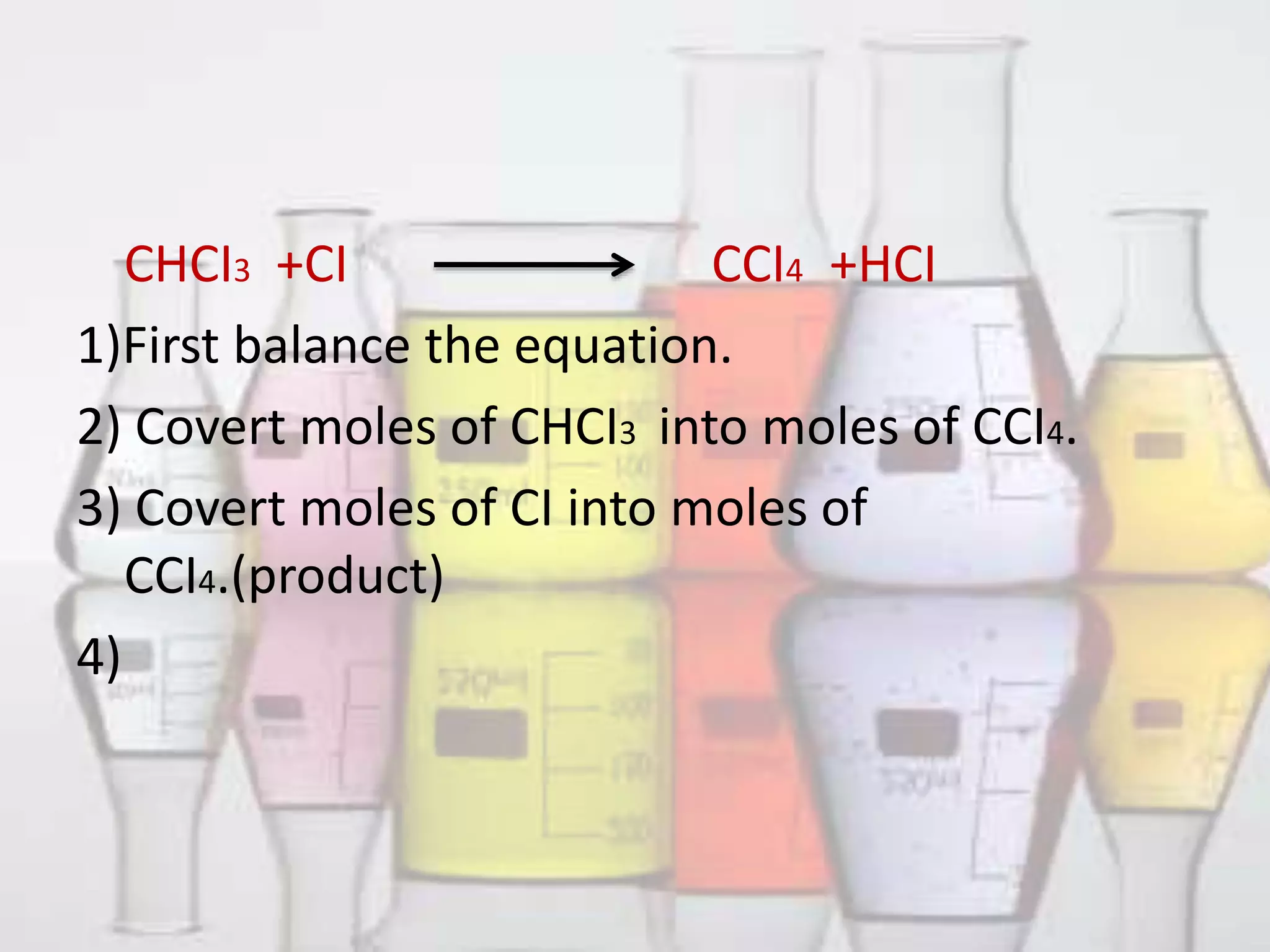
Limiting Reactant And Excess Reactant Pptx Learn how to identify limiting and excess reactants with our step by step stoichiometry guide. calculate theoretical yield easily. master your chemistry exams now!. Limiting reactant is also what prevents a reaction from continuing because there is none left. the limiting reactant may also be referred to as limiting reagent or limiting agent. the reactant that is not used up is referred to as the excess reactant.
Comments are closed.