Integrated Continuous Biomanufacturing Pharma Manufacturing
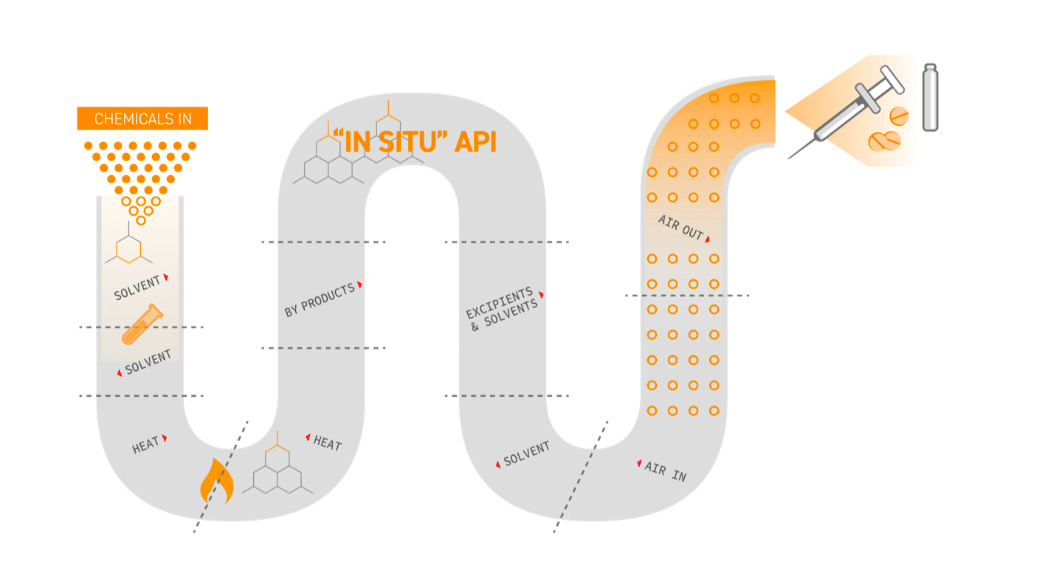
Integrated Continuous Manufacturing Continuus Pharmaceuticals End to end (e2e) continuous biomanufacturing refers to the integrated, uninterrupted process for producing biologics, such as monoclonal antibodies, vaccines, and enzymes, directly from raw materials to the final product. The integrated continuous biomanufacturing (icb) conference series was established in barcelona, spain in 2013, with an international gathering of industry and academic thought leaders across to discuss the promise and challenges of integrated continuous biomanufacturing for biological therapeutics.

301 Moved Permanently The integrated and continuous biomanufacturing (icb) conference is the exclusive worldwide meeting for all interested parties from industry, academia and health authorities to exchange latest advancements in this field that is revolutionizing biopharmaceutical production. This article reviews the integrated and continuous bioprocesses (icb) that are being used, or are soon to be used, in a gmp manufacturing setting for recombinant protein production from mammalian cells, and proposes a common framework. Continuous manufacturing provides a new paradigm for biotherapeutics manufacturing. it has been shown to deliver high productivity (10 15x), lower cost of goods (by 50 75 per cent), and a more consistent product quality. A continuous biomanufacturing platform can process higher lower quantities of a drug as needed and allow manufacturers to respond to changing markets.
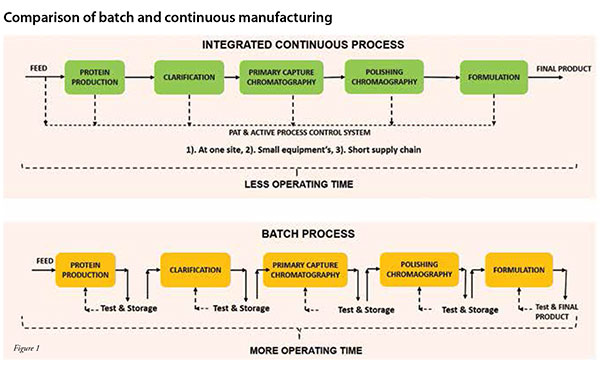
Integrated Continuous Biomanufacturing Pharma Manufacturing Continuous manufacturing provides a new paradigm for biotherapeutics manufacturing. it has been shown to deliver high productivity (10 15x), lower cost of goods (by 50 75 per cent), and a more consistent product quality. A continuous biomanufacturing platform can process higher lower quantities of a drug as needed and allow manufacturers to respond to changing markets. From hybrid platforms to advanced control strategies, this resource showcases how manufacturers are overcoming challenges and building scalable, compliant, and patient focused operations. Discover how advanced biomanufacturing and continuous processing are revolutionizing biopharmaceutical production. transform your biotech processes today!. This review seeks to bridge existing gaps by providing a comprehensive overview of the latest applications of continuous biomanufacturing (cbm), with a particular focus on continuous upstream fermentation and its implications for bioeconomy applications. In addition to significantly increasing manufacturing efficiencies, integrated continuous manufacturing streamlines the complex drug distribution network, reducing logistics costs and expediting the delivery of life saving medicines to patients.
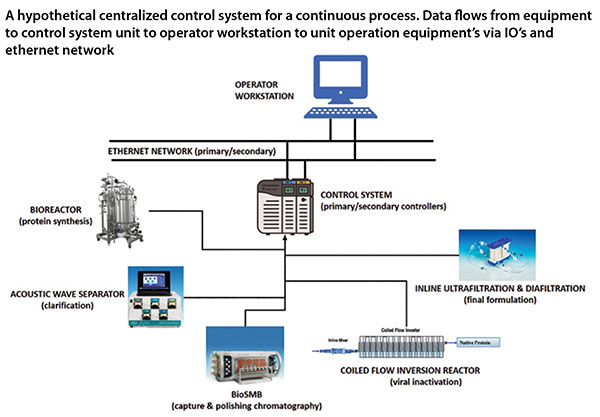
Integrated Continuous Biomanufacturing Pharma Manufacturing From hybrid platforms to advanced control strategies, this resource showcases how manufacturers are overcoming challenges and building scalable, compliant, and patient focused operations. Discover how advanced biomanufacturing and continuous processing are revolutionizing biopharmaceutical production. transform your biotech processes today!. This review seeks to bridge existing gaps by providing a comprehensive overview of the latest applications of continuous biomanufacturing (cbm), with a particular focus on continuous upstream fermentation and its implications for bioeconomy applications. In addition to significantly increasing manufacturing efficiencies, integrated continuous manufacturing streamlines the complex drug distribution network, reducing logistics costs and expediting the delivery of life saving medicines to patients.

Integrated Continuous Biomanufacturing Pharma Manufacturing This review seeks to bridge existing gaps by providing a comprehensive overview of the latest applications of continuous biomanufacturing (cbm), with a particular focus on continuous upstream fermentation and its implications for bioeconomy applications. In addition to significantly increasing manufacturing efficiencies, integrated continuous manufacturing streamlines the complex drug distribution network, reducing logistics costs and expediting the delivery of life saving medicines to patients.

Integrated Continuous Biomanufacturing Pharma Manufacturing
Comments are closed.