Integrated And Continuous Biomanufacturing

301 Moved Permanently This article reviews the integrated and continuous bioprocesses (icb) that are being used, or are soon to be used, in a gmp manufacturing setting for recombinant protein production from mammalian cells, and proposes a common framework. Today, the discussion is about how we can achieve an autonomous bioprocess with integrated continuous biomanufacturing, commercial scale, gmp manufacturing campaigns and the application of continuous bioproduction concepts for production of new modalities.
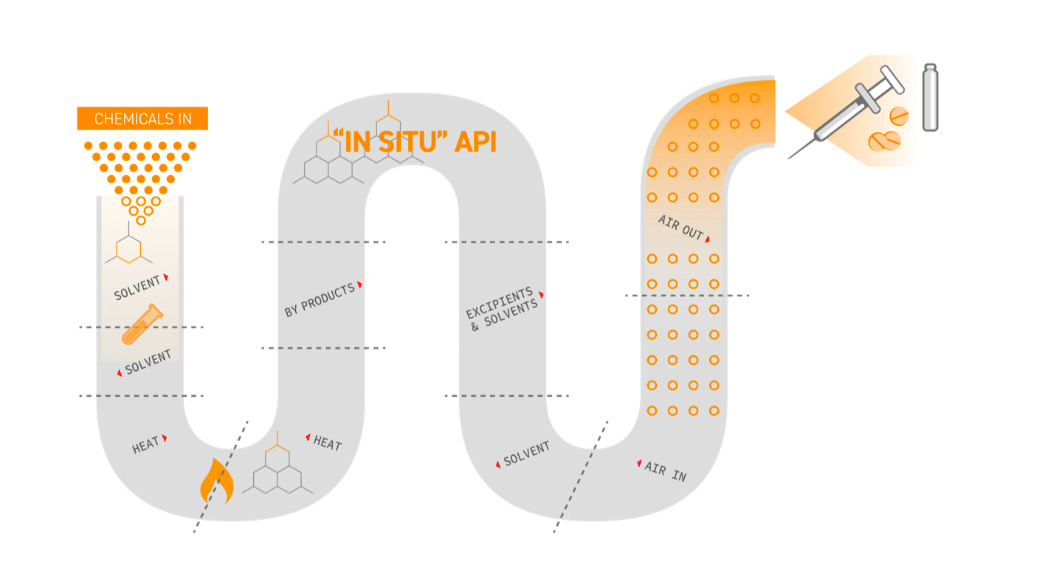
Integrated Continuous Manufacturing Continuus Pharmaceuticals The integrated continuous biomanufacturing (icb) conference series was established in barcelona, spain in 2013, with an international gathering of industry and academic thought leaders across to discuss the promise and challenges of integrated continuous biomanufacturing for biological therapeutics. End to end (e2e) continuous biomanufacturing refers to the integrated, uninterrupted process for producing biologics, such as monoclonal antibodies, vaccines, and enzymes, directly from raw materials to the final product. This review seeks to bridge existing gaps by providing a comprehensive overview of the latest applications of continuous biomanufacturing (cbm), with a particular focus on continuous upstream fermentation and its implications for bioeconomy applications. There is a growing application of integrated and continuous bioprocessing (icb) for manufacturing recombinant protein therapeutics produced from mammalian cells. at first glance, the newly evolved icb has created a vast diversity of platforms.
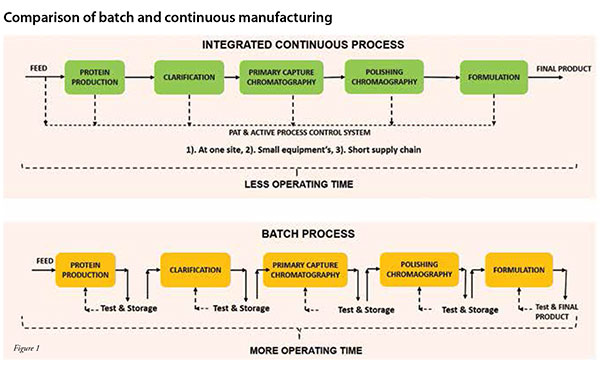
Integrated Continuous Biomanufacturing Pharma Manufacturing This review seeks to bridge existing gaps by providing a comprehensive overview of the latest applications of continuous biomanufacturing (cbm), with a particular focus on continuous upstream fermentation and its implications for bioeconomy applications. There is a growing application of integrated and continuous bioprocessing (icb) for manufacturing recombinant protein therapeutics produced from mammalian cells. at first glance, the newly evolved icb has created a vast diversity of platforms. The regulatory journey for implementation of integrated continuous biomanufacturing for a commercial enzyme replacement therapy product, rebecca berger, phd (abstract). There is a growing application of integrated and continuous bioprocessing (icb) for manufacturing recombinant protein therapeutics produced from mammalian cells. at first glance, the newly. Many biopharmaceutical companies are considering switching from fed batch bioreactors and independent, batch downstream unit operations to integrated and continuous bioprocessing (icb), which uses a mammalian host in continuous perfusion bioreactors and a linked and continuous downstream. Digital twins can enable integrated and continuous biomanufacturing (icb) by providing a digital platform for data integration, analytics, modeling, and decision making across different stages of development and production.
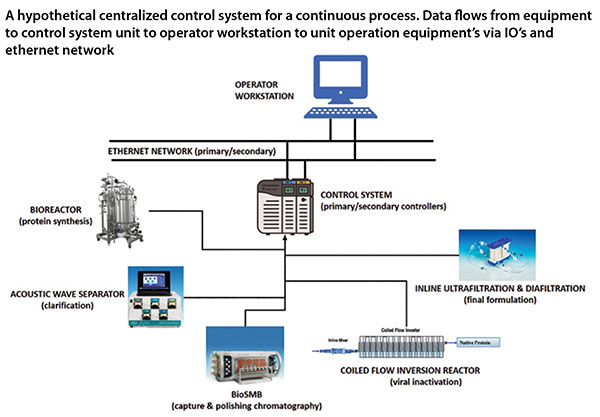
Integrated Continuous Biomanufacturing Pharma Manufacturing The regulatory journey for implementation of integrated continuous biomanufacturing for a commercial enzyme replacement therapy product, rebecca berger, phd (abstract). There is a growing application of integrated and continuous bioprocessing (icb) for manufacturing recombinant protein therapeutics produced from mammalian cells. at first glance, the newly. Many biopharmaceutical companies are considering switching from fed batch bioreactors and independent, batch downstream unit operations to integrated and continuous bioprocessing (icb), which uses a mammalian host in continuous perfusion bioreactors and a linked and continuous downstream. Digital twins can enable integrated and continuous biomanufacturing (icb) by providing a digital platform for data integration, analytics, modeling, and decision making across different stages of development and production.

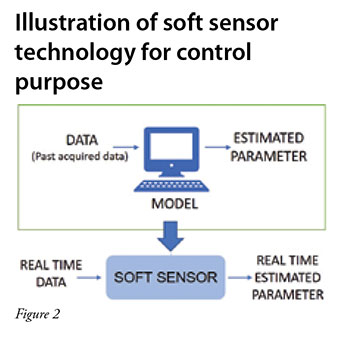
Integrated Continuous Biomanufacturing Pharma Manufacturing Many biopharmaceutical companies are considering switching from fed batch bioreactors and independent, batch downstream unit operations to integrated and continuous bioprocessing (icb), which uses a mammalian host in continuous perfusion bioreactors and a linked and continuous downstream. Digital twins can enable integrated and continuous biomanufacturing (icb) by providing a digital platform for data integration, analytics, modeling, and decision making across different stages of development and production.
Integrated Continuous Biomanufacturing Pharma Manufacturing
Comments are closed.