Injectable Extended Release Hiv Regimen Gets Milestone Fda Approval
Injectable Extended Release Hiv Regimen Gets Milestone Fda Approval Gilead researchers have developed 13 hiv medications, including the first single tablet regimen to treat hiv, the first antiretroviral for pre exposure prophylaxis (prep) to help reduce new hiv infections, and the first long acting injectable hiv treatment medication administered twice yearly. The fda has approved cabotegravir extended release (apretude) as the first long acting injectable prep for hiv prevention. administered every 2 months, it offers a highly effective alternative to daily pills like truvada, with up to 90% risk reduction shown in clinical trials.
Fda Approves First Extended Release Injectable Drug Regimen For Adults The fda has announced its approval of the first injectable extended release regimen for adults who have hiv 1 infections. The approval of doravirine and islatravir marks a new 2 drug, once daily oral option for hiv 1 management, with implications for treatment simplification, adherence, and pharmacist led antiretroviral care. The u.s. food and drug administration (fda) approved yeztugo, a powerful, long acting injection developed by gilead sciences, as a twice yearly preventive treatment against hiv. The tables below list hiv medicines that have been approved by the u.s. food and drug administration (fda) for the treatment or prevention of hiv infection in the united states.
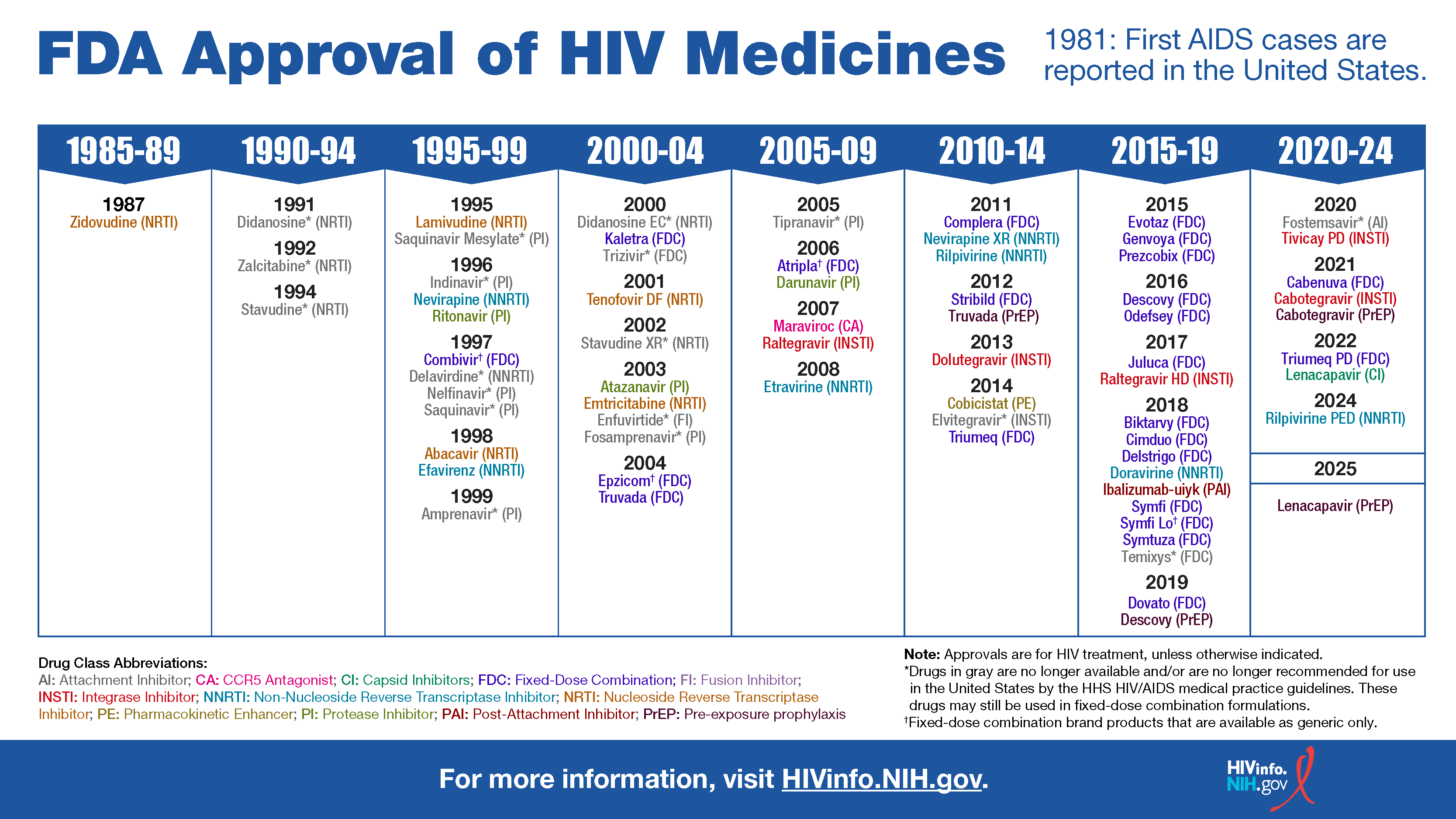
Fda Approval Of Hiv Medicines The u.s. food and drug administration (fda) approved yeztugo, a powerful, long acting injection developed by gilead sciences, as a twice yearly preventive treatment against hiv. The tables below list hiv medicines that have been approved by the u.s. food and drug administration (fda) for the treatment or prevention of hiv infection in the united states. Rahway, n.j. (business wire) merck (nyse: mrk), known as msd outside of the united states and canada, announced today that the u.s. food and drug administration (fda) approved idvynso™, a new, two drug single tablet regimen of 100 mg doravirine and 0.25 mg islatravir, for the treatment of hiv 1 infection in adults to replace the current antiretroviral regimen in those who are. Individuals must be tested for hiv 1 infection prior to initiating apretude or oral cabotegravir, and with each subsequent injection of apretude, using a test approved or cleared by the fda. April 21 (reuters) the u.s. food and drug administration has approved merck's once daily, oral, combination regimen for hiv infections, the drugmaker said on tuesday, giving patients. Cabenuva is fda approved for patients who are virally suppressed on a stable oral arv regimen, with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine.
Comments are closed.