Ich Q2 Analytical Method Validation

Ich Q2r2 Guideline Validation Analytical Procedures Step 5 Revision 1 Analytical procedure validation forms a part of the analytical procedure lifecycle, as described within ich q14 analytical procedure development. ich q2(r2) provides guidance on selection and evaluation of the various validation tests for analytical procedures. This guideline presents a discussion of elements for consideration during the validation of analytical procedures included as part of registration applications submitted within the ich member regulatory authorities.
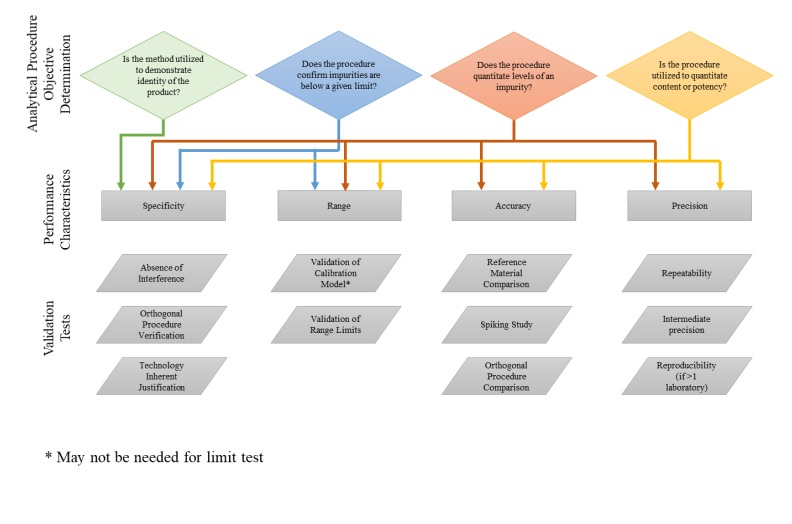
Ich Q2 R1 Pharmasciences Fda guidance (2015): “analytical procedures and methods validation for drugs and biologics” provides detail on fda’s view of method validation, essentially harmonizing with ich q2 concepts but emphasizing thorough documentation and modern quality systems. This review provides a detailed comparative analysis between ich q2 (r1) and the new q2 (r2) guideline, highlighting key changes in validation parameters such as specificity, linearity, accuracy, precision, limit of detection (lod), and quantitation (loq). A validation study is designed to provide sufficient evidence that the analytical procedure meets its objectives. these objectives are described with a suitable set of performance. This document presents a discussion of the characteristics for consideration during the validation of the analytical procedures included as part of registration applications submitted within the ec, japan and usa.

Ich Q2 Analytical Method Validation Pptx A validation study is designed to provide sufficient evidence that the analytical procedure meets its objectives. these objectives are described with a suitable set of performance. This document presents a discussion of the characteristics for consideration during the validation of the analytical procedures included as part of registration applications submitted within the ec, japan and usa. Provides guidance for validating multivariate model based methods (e.g., nir, raman) and hyphenated techniques (e.g., lc–ms), including model maintenance, periodic verification, and revalidation when models are updated. Analytical method validation ensures reliable and compliant data throughout drug development. with the introduction of ich q2 (r2) and q14, expectations around method development, validation and lifecycle management have significantly evolved. analytical method validation is a critical and a widely studied process in industries such as pharmaceuticals, food safety, biotechnology, and. Ich q2 (r2) builds upon the earlier q2 (r1) guidance, clarifying the principles behind analytical method validation, defining the studies, performance characteristics, and acceptance criteria needed to demonstrate a method is fit for its intended purpose. Ich q2 (r2) presents a discussion of the characteristics to be considered during the validation of analytical procedures included as part of registration documentation within the ich region.

Ich Q2 Analytical Method Validation Pptx Provides guidance for validating multivariate model based methods (e.g., nir, raman) and hyphenated techniques (e.g., lc–ms), including model maintenance, periodic verification, and revalidation when models are updated. Analytical method validation ensures reliable and compliant data throughout drug development. with the introduction of ich q2 (r2) and q14, expectations around method development, validation and lifecycle management have significantly evolved. analytical method validation is a critical and a widely studied process in industries such as pharmaceuticals, food safety, biotechnology, and. Ich q2 (r2) builds upon the earlier q2 (r1) guidance, clarifying the principles behind analytical method validation, defining the studies, performance characteristics, and acceptance criteria needed to demonstrate a method is fit for its intended purpose. Ich q2 (r2) presents a discussion of the characteristics to be considered during the validation of analytical procedures included as part of registration documentation within the ich region.

Ich Q2 Analytical Method Validation Pptx Ich q2 (r2) builds upon the earlier q2 (r1) guidance, clarifying the principles behind analytical method validation, defining the studies, performance characteristics, and acceptance criteria needed to demonstrate a method is fit for its intended purpose. Ich q2 (r2) presents a discussion of the characteristics to be considered during the validation of analytical procedures included as part of registration documentation within the ich region.
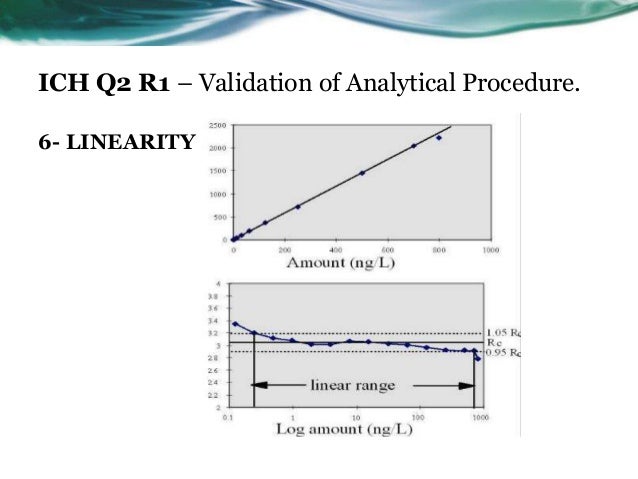
Ich Q2 Analytical Method Validation
Comments are closed.