Hydrogen Sulfide H2s Gas Vs Sulfur Dioxide Key Differences Explained

Hydrogen Sulfide H2s Gas Vs Sulfur Dioxide Key Differences Explained In this article, we will dive into the key differences between hydrogen sulfide and sulfur dioxide, honing in on their chemical characteristics, sources, health impacts, and environmental roles. H2s, also known as hydrogen sulfide, is a chemical compound composed of two hydrogen atoms bonded to a sulfur atom. it is a colorless gas with a distinct odor of rotten eggs. on the other hand, so2, or sulfur dioxide, consists of one sulfur atom bonded to two oxygen atoms.

Hydrogen Sulfide H2s Gas Sulfur Dioxide So2 Gas Hydrogen Chloride Hcl Hydrogen sulfide (h2s) and sulfur dioxide (so2) are both inorganic gaseous compounds containing sulfur, each possessing distinct properties and playing unique roles in various natural and industrial processes. The key difference between h2s and so2 is that h2s has the smell of a rotten egg, whereas so2 has the smell of a burnt match. both h2s and so2 are gaseous compounds at room temperature. Hydrogen sulfide blocks the body’s ability to use oxygen, whereas sulfur dioxide inflames the respiratory system and forms acid particles that surely damage lungs and the environment. H2s is a colorless, flammable, and highly toxic gas with a "rotten egg" odor at low concentrations. so2 is also a colorless gas, but it has a pungent, irritating odor similar to burnt matches.
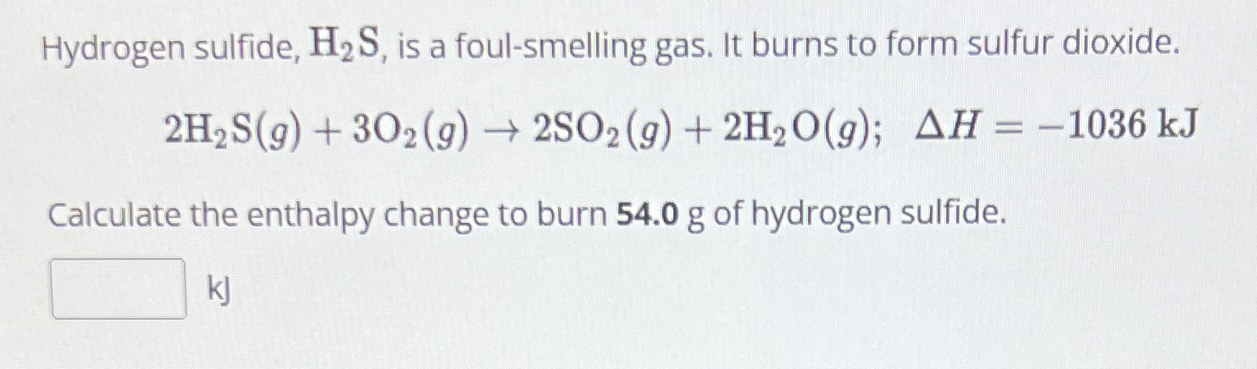
Solved Hydrogen Sulfide H2s ï Is A Foul Smelling Gas It Chegg Hydrogen sulfide blocks the body’s ability to use oxygen, whereas sulfur dioxide inflames the respiratory system and forms acid particles that surely damage lungs and the environment. H2s is a colorless, flammable, and highly toxic gas with a "rotten egg" odor at low concentrations. so2 is also a colorless gas, but it has a pungent, irritating odor similar to burnt matches. Hydrogen sulfide (h2 s), like carbon dioxide, is a type of acid gas that is an undesirable byproduct in the oil and gas industry. h 2 s is a colorless, poisonous, flammable, and toxic gas with the odor of rotten eggs. Hydrogen sulfide burns in oxygen with a blue flame to form sulfur dioxide (so2) and water: 2 h2s 3 o2 → 2 so2 2 h2o. if an excess of oxygen is present, sulfur trioxide (so3) is formed, which quickly hydrates to sulfuric acid: h2s 2 o2 → h2so4. Perhaps the shortest and easiest explanations given these differences is that at lower levels sulfur dioxide can be described as a more corrosive irritant—especially when the lungs are concerned—and at moderately higher levels hydrogen sulfide can be described as an effective neurotoxin. Even if single hydrogen bonds transiently form between h 2 s and h 2 o, the h 2 s molecule cannot sustain any further hydrogen bonds, whereas the h 2 o molecules prefer having four hydrogen bonds.
Comments are closed.