Hydrogen Atomic Structure

Hydrogen Atomic Structure A hydrogen atom is an atom of the chemical element hydrogen. the electrically neutral hydrogen atom contains a single positively charged proton in the nucleus, and a single negatively charged electron bound to the nucleus by the coulomb force. The hydrogen atom consists of a single negatively charged electron that moves about a positively charged proton (figure 8 2 1). in bohr’s model, the electron is pulled around the proton in a perfectly circular orbit by an attractive coulomb force.

Hydrogen Atomic Structure The hydrogen atom consists of a single negatively charged electron that moves about a positively charged proton ((figure)). in bohr’s model, the electron is pulled around the proton in a perfectly circular orbit by an attractive coulomb force. Learn about the hydrogen atom, the simplest and most abundant atom in the universe, and its role in quantum mechanics and energy research. explore its structure, energy levels, spectral lines, and applications in astronomy, technology, and education. Learn how to draw and interpret the hydrogen atomic structure diagram, which shows the nucleus, electron, and energy levels of the simplest element. explore the basics, properties, and applications of hydrogen, as well as its isotopes and role in the universe. What does the bohr model explain? the bohr model could account for the series of discrete wavelengths in the emission spectrum of hydrogen. niels bohr proposed that light radiated from hydrogen atoms only when an electron made a transition from an outer orbit to one closer to the nucleus.
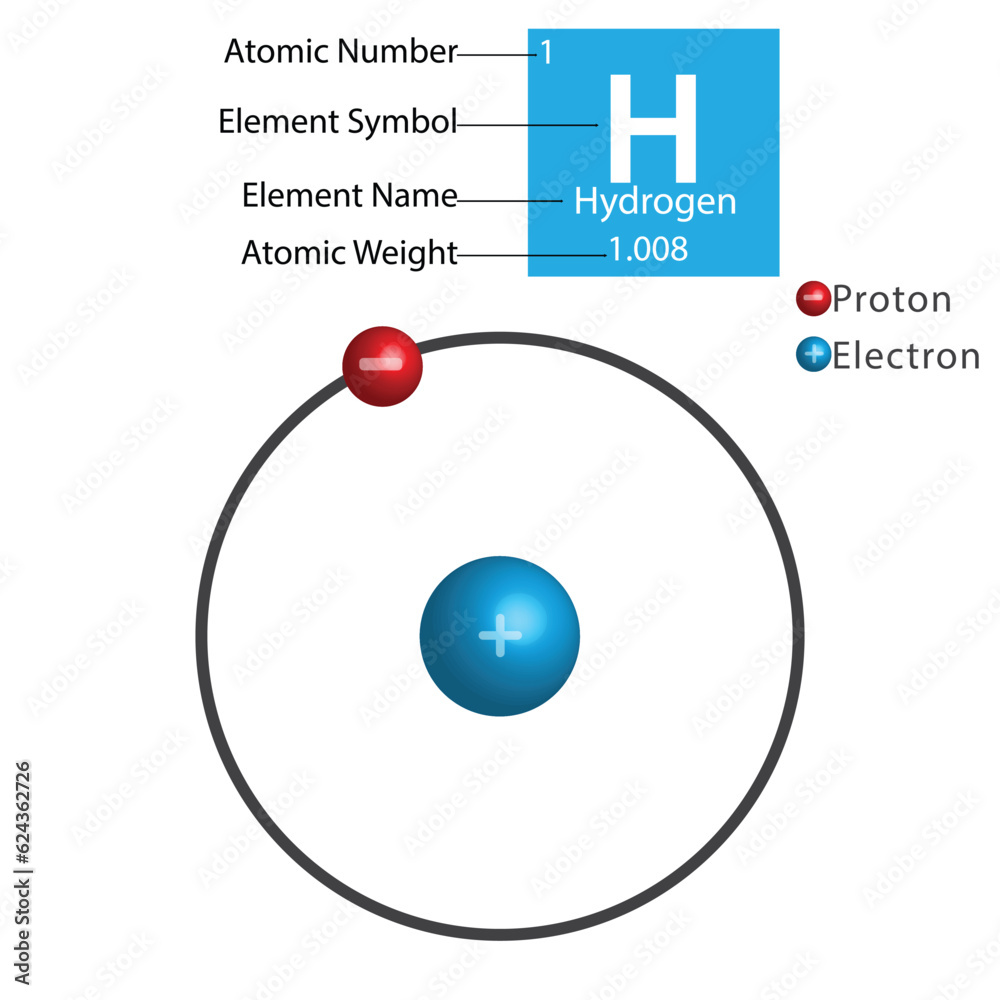
What Is The Atomic Structure Of A Hydrogen Atom Learn how to draw and interpret the hydrogen atomic structure diagram, which shows the nucleus, electron, and energy levels of the simplest element. explore the basics, properties, and applications of hydrogen, as well as its isotopes and role in the universe. What does the bohr model explain? the bohr model could account for the series of discrete wavelengths in the emission spectrum of hydrogen. niels bohr proposed that light radiated from hydrogen atoms only when an electron made a transition from an outer orbit to one closer to the nucleus. Visualize and understand the atomic structure of hydrogen, including electron shells and configuration. This interactive comparison shows the atomic structures for hydrogen and helium. Learn about the basic particles and properties of atoms, such as protons, neutrons, electrons, atomic number, mass number, and isotopes. find out how hydrogen atoms differ from other atoms and how they are composed of one proton and one electron. First, it's simple. it's just an electron bound by the pull of a proton. second, other atoms and molecules can be build upwards from the lessons taught by hydrogen. atomic and molecular physics thus start with an understanding of hydrogen.
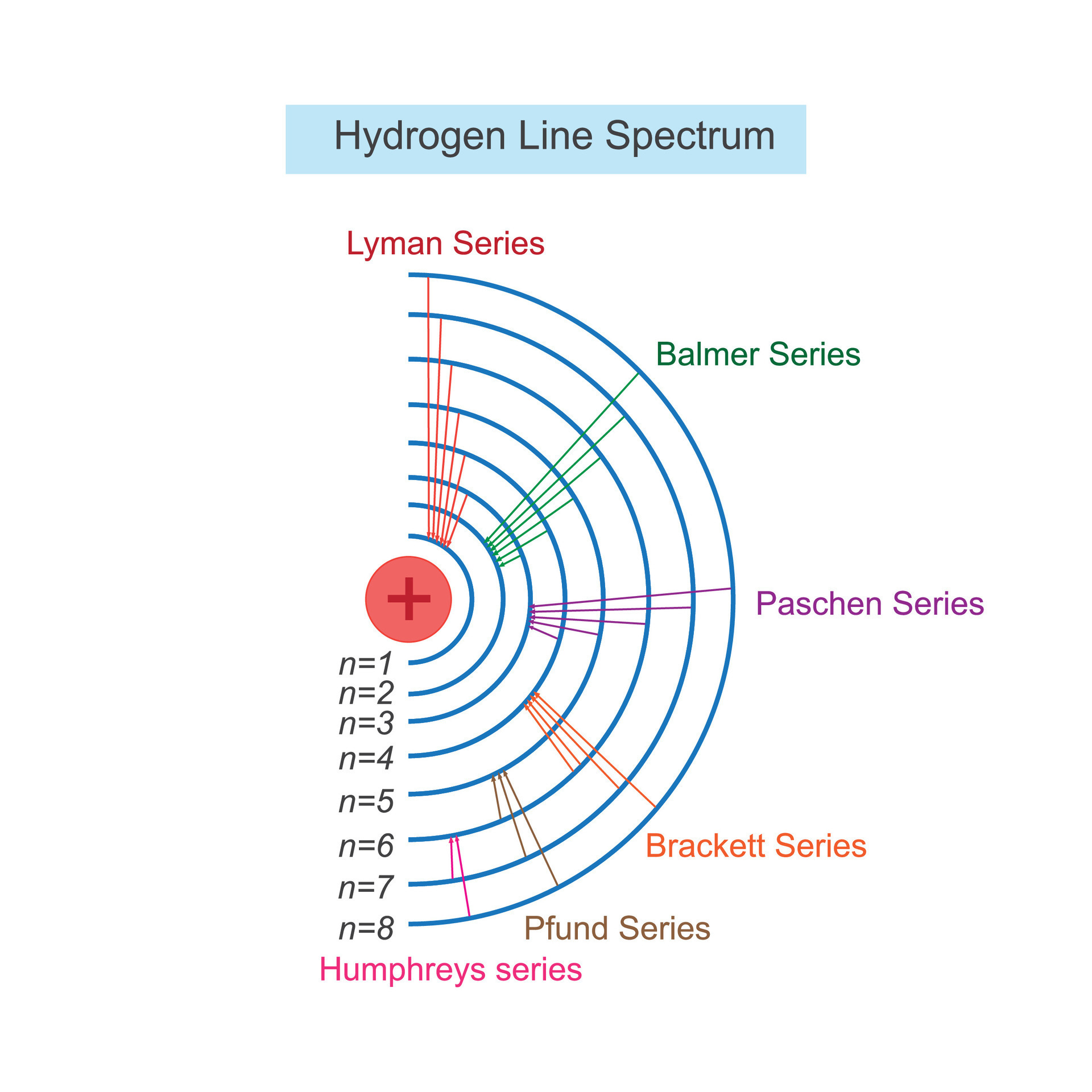
Atomic Structure Illustration Line Spectrum Of Hydrogen Atom Bohr Visualize and understand the atomic structure of hydrogen, including electron shells and configuration. This interactive comparison shows the atomic structures for hydrogen and helium. Learn about the basic particles and properties of atoms, such as protons, neutrons, electrons, atomic number, mass number, and isotopes. find out how hydrogen atoms differ from other atoms and how they are composed of one proton and one electron. First, it's simple. it's just an electron bound by the pull of a proton. second, other atoms and molecules can be build upwards from the lessons taught by hydrogen. atomic and molecular physics thus start with an understanding of hydrogen.
Comments are closed.