Hydrogen Atom Wavefunctions
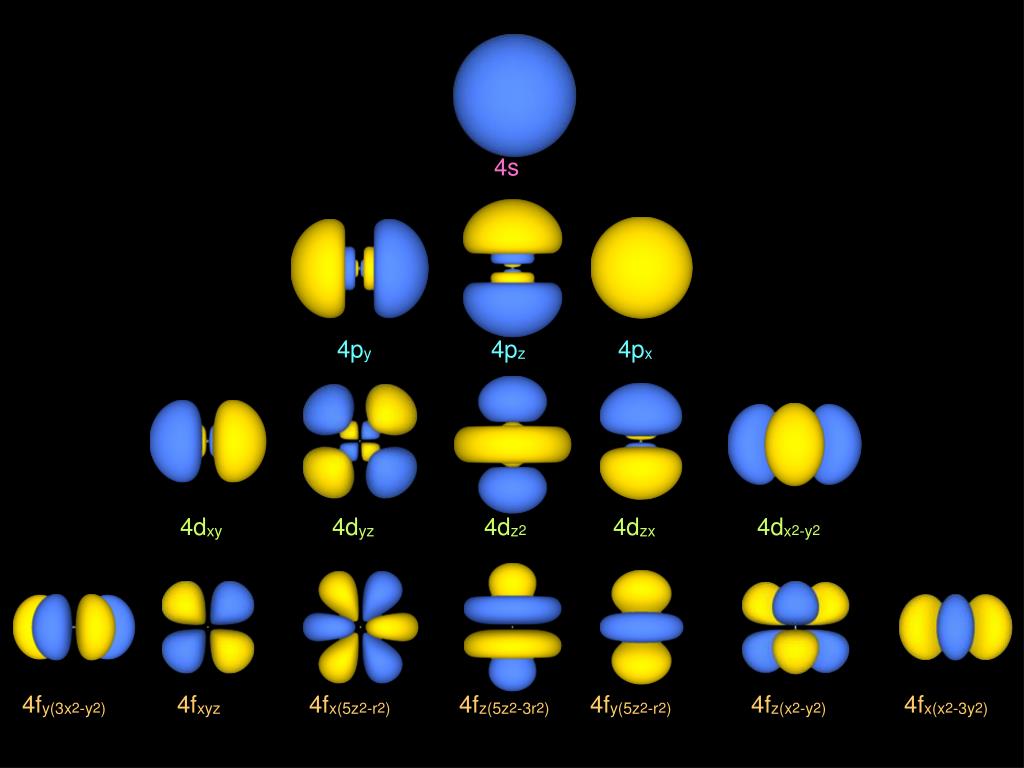
Ppt Atomic Orbital Hydrogen Atom Wave Function 47 Off Source: beiser, a., perspectives of modern physics, mcgraw hill, 1969. table 9.1. source: beiser, a., perspectives of modern physics, mcgraw hill, 1969. table 9.1. Other one electron systems have electronic states analogous to those for the hydrogen atom, and inclusion of the charge on the nucleus allows the same wavefunctions to be used for all one electron systems.
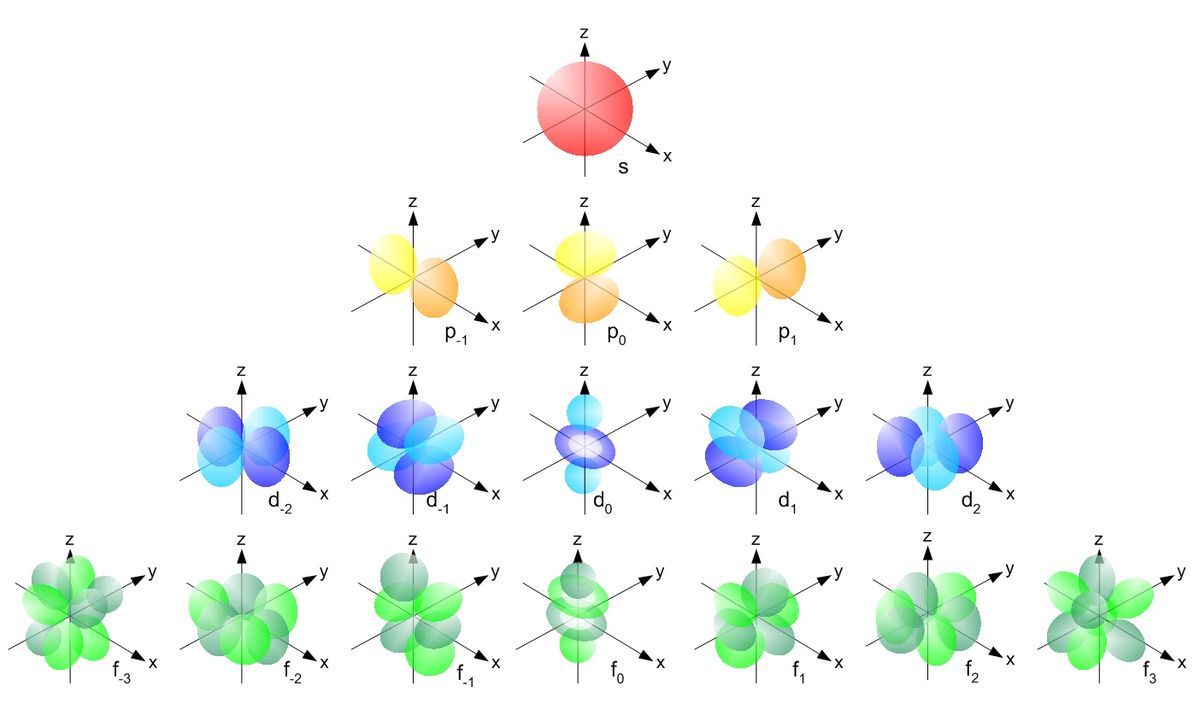
Hydrogen Atom Brilliant Math Science Wiki A hydrogen atom is an atom of the chemical element hydrogen. the electrically neutral hydrogen atom contains a single positively charged proton in the nucleus, and a single negatively charged electron bound to the nucleus by the coulomb force. The dutch physicist pieter zeeman showed the spectral lines emitted by atoms in a magnetic field split into multiple energy levels. it is called the zeeman effect. In this session, the probability of finding an electron at a particular distance from the nucleus is discussed. the concept of wavefunctions (orbitals) is introduced, and applications of electron spin are described. For hydrogen, the simplest atom, it describes the electron's behavior using a wavefunction. this function contains all the info about the electron's position and energy in the atom.
Hydrogen Atom Orbitals Prompts Stable Diffusion Online In this session, the probability of finding an electron at a particular distance from the nucleus is discussed. the concept of wavefunctions (orbitals) is introduced, and applications of electron spin are described. For hydrogen, the simplest atom, it describes the electron's behavior using a wavefunction. this function contains all the info about the electron's position and energy in the atom. The wavefunctions for the hydrogen atom depend upon the three variables r, θ, and ϕ and the three quantum numbers n, l, and m l. the variables give the position of the electron relative to the nucleus in spherical coordinates. Most textbooks treat the h atom as an exactly solved problem. the solution is based on standard techniques for solving differential equations and the solutions are expressed in terms of named and tabulated special function. What is the hydrogen atom wave function? the hydrogen atom wave function describes the quantum mechanical behavior of electrons in a hydrogen atom. unlike classical physics, electrons do not move in fixed orbits but exist as probability clouds around the nucleus. But after schrödinger, the universe suddenly became more abstract; it is now—at least in schrödinger’s treatment of the hydrogen atom—made up of wave functions, which we really don’t understand.

Hydrogen Atom The Energy Of The Electron Of The Hydrogen Atom In The The wavefunctions for the hydrogen atom depend upon the three variables r, θ, and ϕ and the three quantum numbers n, l, and m l. the variables give the position of the electron relative to the nucleus in spherical coordinates. Most textbooks treat the h atom as an exactly solved problem. the solution is based on standard techniques for solving differential equations and the solutions are expressed in terms of named and tabulated special function. What is the hydrogen atom wave function? the hydrogen atom wave function describes the quantum mechanical behavior of electrons in a hydrogen atom. unlike classical physics, electrons do not move in fixed orbits but exist as probability clouds around the nucleus. But after schrödinger, the universe suddenly became more abstract; it is now—at least in schrödinger’s treatment of the hydrogen atom—made up of wave functions, which we really don’t understand.
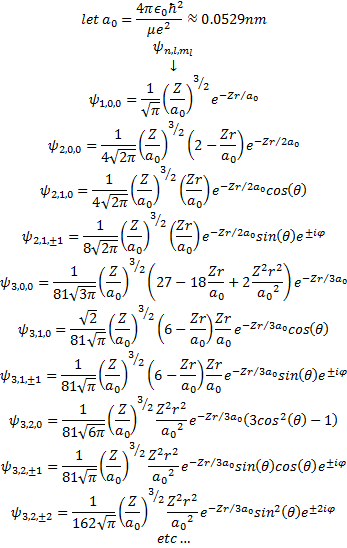
Hydrogen Atom Wavefunctions What is the hydrogen atom wave function? the hydrogen atom wave function describes the quantum mechanical behavior of electrons in a hydrogen atom. unlike classical physics, electrons do not move in fixed orbits but exist as probability clouds around the nucleus. But after schrödinger, the universe suddenly became more abstract; it is now—at least in schrödinger’s treatment of the hydrogen atom—made up of wave functions, which we really don’t understand.
Comments are closed.