How To Recrystallize A Solid

Recrystallization Dissolve Your Solid And Precipitate Your Crystals Recrystallization helps purify a solid using a solvent and different solubility at various temperatures. heat the mixture to dissolve the solid, then cool it to form pure crystals. vacuum filtration separates and dries the purified crystals from the remaining liquid. Recrystallization, also known as fractional crystallization, is a procedure for purifying an impure compound in a solvent. the method of purification is based on the principle that the solubility of most solids increases with increased temperature.

Comparison Of Recrystallization Transfer the material to a 50 ml erlenmeyer flask equipped with a stir bar. add about 20 ml of the solvent (determined in part i) and heat to boiling on a stir hot plate. remove any insoluble impurities by filtration, and recrystallize your product— see two solvent recrystallization guide. Recrystallization is a method employed to purify solid compounds. it involves dissolving the impure solid in a heated solvent until the solution reaches saturation, followed by gradual cooling. as the solution cools, pure crystals form while impurities are left behind. Scientists use recrystallization to purify solids, typically products, from different chemical reactions. the process involves dissolving a solid into solution, then allowing the dissolved substance to gradually crystallize. During recrystallization, an impure solid compound is dissolved in a hot liquid until the solution is saturated, and then the liquid is allowed to cool.2 the compound should then form relatively pure crystals.

Chemical Laboratory Techniques Recrystallization Youtube Scientists use recrystallization to purify solids, typically products, from different chemical reactions. the process involves dissolving a solid into solution, then allowing the dissolved substance to gradually crystallize. During recrystallization, an impure solid compound is dissolved in a hot liquid until the solution is saturated, and then the liquid is allowed to cool.2 the compound should then form relatively pure crystals. In principle, the supernatant solution can be decanted from the solid. however, a loss of the solid as well as an incomplete removal of the liquid, which might contain the impurities, will take place. a better technique to separate the solid from the liquid is by filtration. By means of a technique called recrystallization, many solids can be purified using pure solvents or solvent mixtures. recrystallization is based on the different solubility of a solid substance in a solvent at room temperature or when the solvent is hot. Recrystallization is a widely used technique in chemistry aimed at purifying solid substances. this process involves dissolving the substance along with its impurities in a suitable solvent at elevated temperatures. Recrystallization is a laboratory technique for purifying solids. the key features of this technique is causing a solid to go into solution, and then gradually allowing the dissolved solid to crystallize.
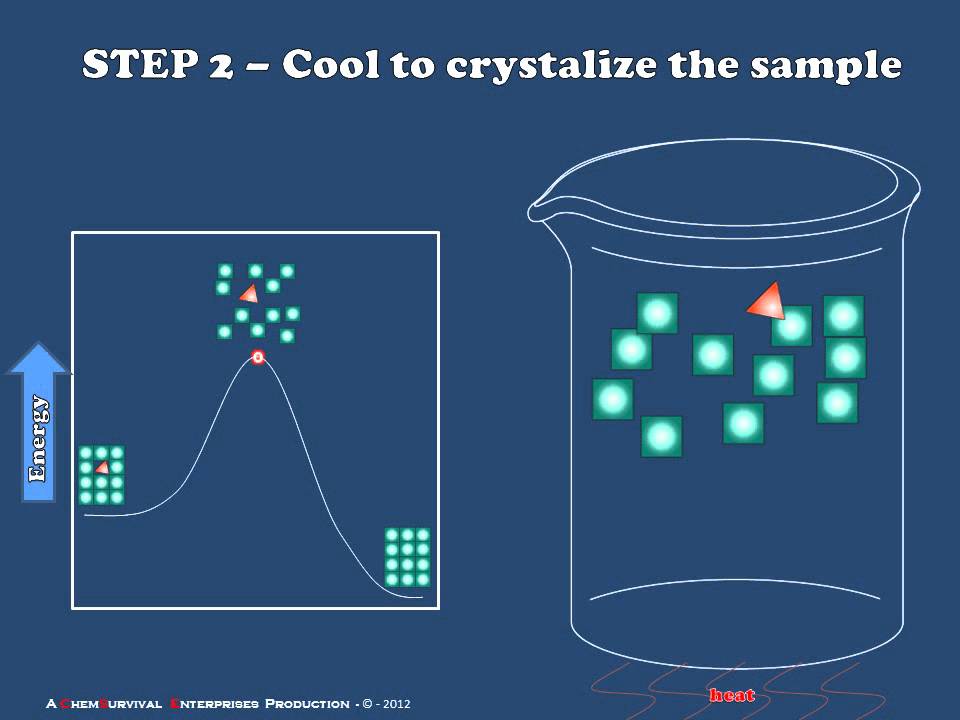
Introduction To Recrystallization Youtube In principle, the supernatant solution can be decanted from the solid. however, a loss of the solid as well as an incomplete removal of the liquid, which might contain the impurities, will take place. a better technique to separate the solid from the liquid is by filtration. By means of a technique called recrystallization, many solids can be purified using pure solvents or solvent mixtures. recrystallization is based on the different solubility of a solid substance in a solvent at room temperature or when the solvent is hot. Recrystallization is a widely used technique in chemistry aimed at purifying solid substances. this process involves dissolving the substance along with its impurities in a suitable solvent at elevated temperatures. Recrystallization is a laboratory technique for purifying solids. the key features of this technique is causing a solid to go into solution, and then gradually allowing the dissolved solid to crystallize.
Comments are closed.