How Indicators Work
Indicators Acids And Bases Indicators are substances whose solutions change color due to changes in ph. these are called acid base indicators. they are usually weak acids or bases, but their conjugate base or acid forms have different colors due to differences in their absorption spectra. During the webinar, we will explore what technical analysis means, what technical indicators are, and how they may be applied. we will take a look at the main types of technical indicators.
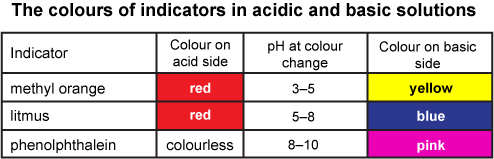
Indicators Acids And Bases Indicators are substances that change colour when they are added to acidic or alkaline solutions. litmus, phenolphthalein, and methyl orange are all indicators that are commonly used in the laboratory. Chemical indicators are specialized substances that provide a visible sign, typically a change in color, to reveal specific chemical conditions within a solution. these compounds are invaluable tools in chemistry, allowing for quick and straightforward assessment of a substance’s properties. In this post, we’ll explore what indicators are, how they work, their different types, and why they are so useful in science and daily life. what are indicators? an indicator is a substance that shows a visible change—usually a color change —when it is placed in a solution. An indicator is a weak acid that ionizes within a known ph range, usually about 2 ph units. we can represent the protonated form of the indicator molecule as hin and the deprotonated form as in −.
.png)
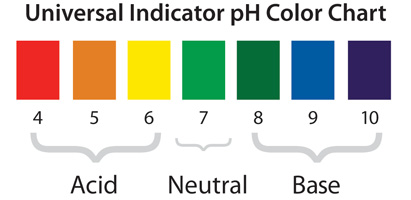
How Indicators Work In this post, we’ll explore what indicators are, how they work, their different types, and why they are so useful in science and daily life. what are indicators? an indicator is a substance that shows a visible change—usually a color change —when it is placed in a solution. An indicator is a weak acid that ionizes within a known ph range, usually about 2 ph units. we can represent the protonated form of the indicator molecule as hin and the deprotonated form as in −. Definition: indicators are substances that change color in response to a change in ph. purpose: used to determine the endpoint of a titration and to gauge the ph of a solution. Indicators work in chemical reactions by changing their properties, usually colour, in response to changes in the chemical environment. this change is often due to the interaction between the indicator and the hydrogen or hydroxide ions in the solution. How does that work? an indicator is a weak acid that ionizes within a known ph range, usually about 2 ph units. we can represent the protonated form of the indicator molecule as hin and the deprotonated form as in a. the following equilibrium exists for the indicator: hin (a q) ⇌ h a (a q) in a (a q). Indicators in chemistry are substances that undergo a readily observable change, typically a color change, in response to changes in the chemical environment around them. this response is usually triggered by alterations in ph, oxidation reduction potential (redox), or the presence of specific ions or compounds.
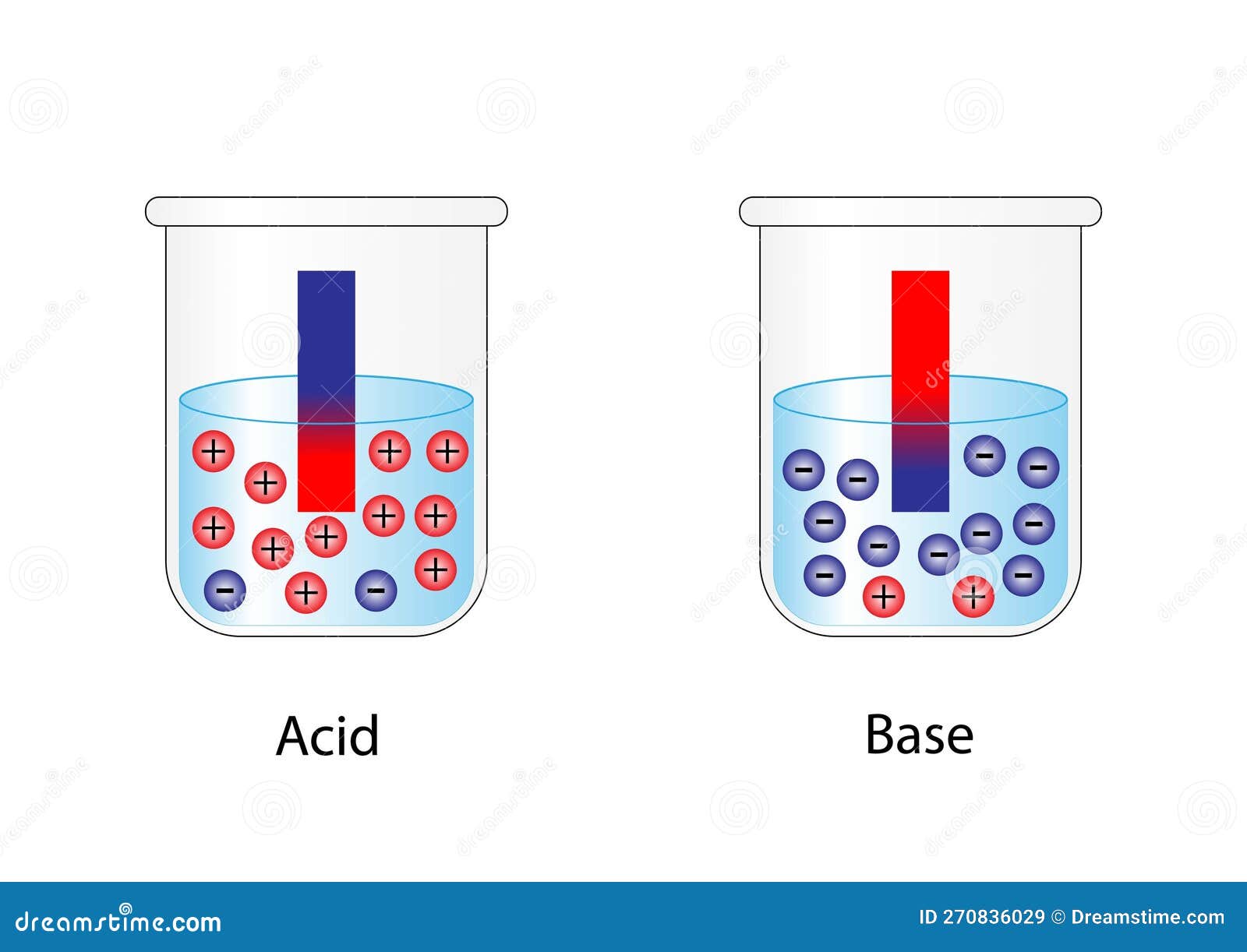
Chemical Indicators Definition Types And Examples 56 Off Definition: indicators are substances that change color in response to a change in ph. purpose: used to determine the endpoint of a titration and to gauge the ph of a solution. Indicators work in chemical reactions by changing their properties, usually colour, in response to changes in the chemical environment. this change is often due to the interaction between the indicator and the hydrogen or hydroxide ions in the solution. How does that work? an indicator is a weak acid that ionizes within a known ph range, usually about 2 ph units. we can represent the protonated form of the indicator molecule as hin and the deprotonated form as in a. the following equilibrium exists for the indicator: hin (a q) ⇌ h a (a q) in a (a q). Indicators in chemistry are substances that undergo a readily observable change, typically a color change, in response to changes in the chemical environment around them. this response is usually triggered by alterations in ph, oxidation reduction potential (redox), or the presence of specific ions or compounds.

Acid And Base Indicators Chemtalk How does that work? an indicator is a weak acid that ionizes within a known ph range, usually about 2 ph units. we can represent the protonated form of the indicator molecule as hin and the deprotonated form as in a. the following equilibrium exists for the indicator: hin (a q) ⇌ h a (a q) in a (a q). Indicators in chemistry are substances that undergo a readily observable change, typically a color change, in response to changes in the chemical environment around them. this response is usually triggered by alterations in ph, oxidation reduction potential (redox), or the presence of specific ions or compounds.
Comments are closed.