How Ftc Compliance Works Labeling Your Dietary Supplements

Labeling Dietary Supplements Pdf Nutraceutical Dietary Supplements Marketers of health related products, including dietary supplements, should be familiar with the requirements under both fda law and ftc law that labeling and advertising claims be truthful, not misleading, and substantiated. While the fda governs product labeling, the ftc oversees dietary supplement advertising law, ensuring that promotional materials are truthful, substantiated, and not deceptive.

Labeling Dietary Supplements Pdf Master dietary supplement marketing regulations with this comprehensive guide. learn fda, ftc rules, compliant health claims, labeling & avoid costly mistakes. Ftc and fda compliance checklist for supplement merchants. learn how labeling, claims, subscriptions, and reviews affect underwriting and payment stability. The u.s. food and drug administration and ftc share responsibility for the oversight of dietary supplements and related promotion. fda is generally responsible for labeling, safety and quality, while ftc is generally responsible for advertising. On december 20, 2022, the federal trade commission (“ftc”) issued a new guidance document entitled health products compliance guidance. the publication updates and replaces ftc’s 1998 document, dietary supplements: an advertising guide for industry.
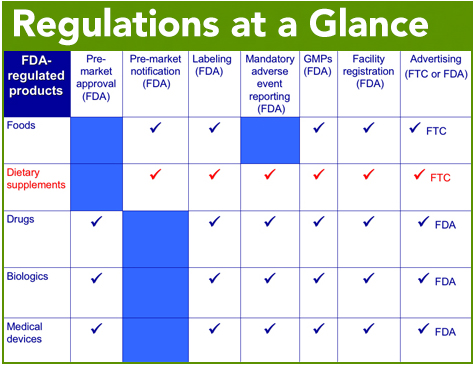
Dietary Supplements Are Regulated By Fda And Ftc Council For The u.s. food and drug administration and ftc share responsibility for the oversight of dietary supplements and related promotion. fda is generally responsible for labeling, safety and quality, while ftc is generally responsible for advertising. On december 20, 2022, the federal trade commission (“ftc”) issued a new guidance document entitled health products compliance guidance. the publication updates and replaces ftc’s 1998 document, dietary supplements: an advertising guide for industry. One major revision is to extend the guidance covering dietary supplements to all health related products. the revised guide also reflects updates to other ftc guidance documents, including the guidance on endorsements and testimonials and the enforcement policy statement on homeopathic drugs. You must list dietary ingredients without rdis or drvs in the "supplement facts" panel for dietary supplements. you are not permitted to list these ingredients in the "nutrition facts". Given the dramatic increase in the volume and variety of dietary supplement advertising in recent years, ftc staff is issuing this guide to clarify how long standing ftc policies and enforcement practices relate to dietary supplement advertising. By following this step by step tutorial, you will be equipped to navigate the complexities of compliance and minimize the risks of enforcement actions.
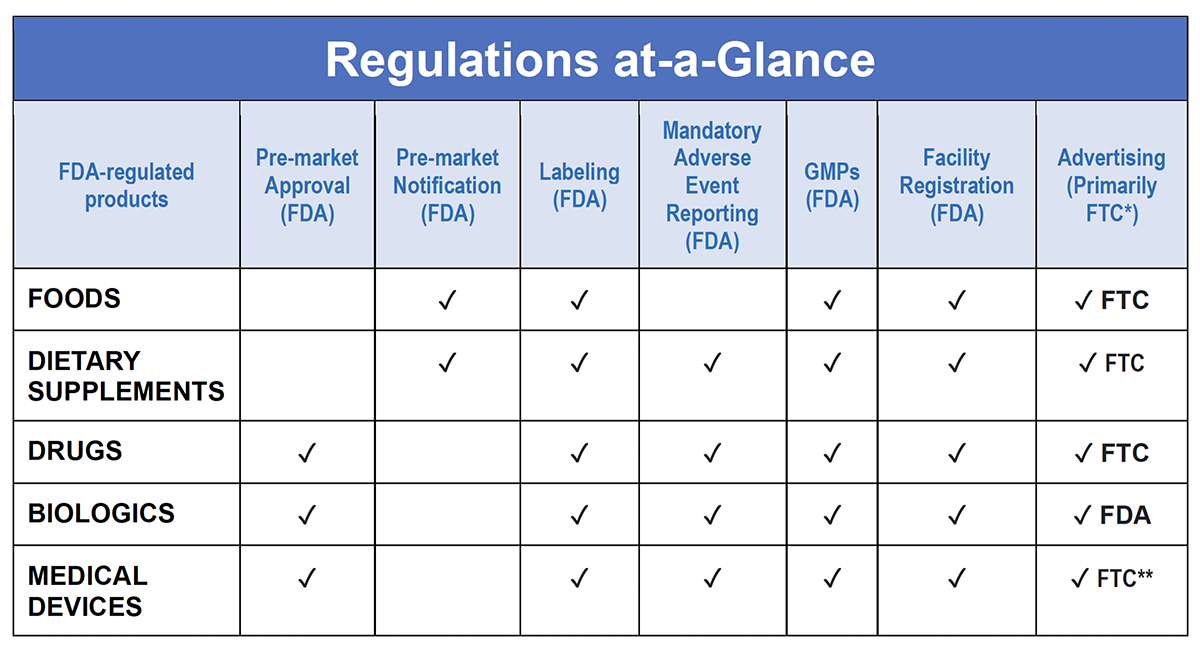
Dietary Supplements Are Regulated By Fda And Ftc Council For One major revision is to extend the guidance covering dietary supplements to all health related products. the revised guide also reflects updates to other ftc guidance documents, including the guidance on endorsements and testimonials and the enforcement policy statement on homeopathic drugs. You must list dietary ingredients without rdis or drvs in the "supplement facts" panel for dietary supplements. you are not permitted to list these ingredients in the "nutrition facts". Given the dramatic increase in the volume and variety of dietary supplement advertising in recent years, ftc staff is issuing this guide to clarify how long standing ftc policies and enforcement practices relate to dietary supplement advertising. By following this step by step tutorial, you will be equipped to navigate the complexities of compliance and minimize the risks of enforcement actions.
Comments are closed.