Hope As Fda Approves Injectable Drug For Resistant Hiv Daily Nation

Hope As Fda Approves Injectable Drug For Resistant Hiv Daily Nation Yeztugo had already been approved in 2022 for multidrug resistant infections under the name sunlenca in combination with other antiretrovirals, with a price set at $42,250 annually. the. The us food and drug administration (fda) has approved lenacapavir, an injectable drug that offers long lasting protection against hiv infection. administered once every six months, this new treatment marks a major advancement in expanding prevention options for people at risk of hiv infection.

Fda Approves New Hiv Drug For Adults With Limited Treatment Options Who welcomes the approval by the united states food and drug administration (fda) of injectable lenacapavir for hiv prevention. administered just twice a year, lenacapavir offers sustained protection and adds to the growing range of hiv prevention options. A drug with the potential to drastically curb the hiv epidemic just cleared its first regulatory hurdle. on wednesday, the food and drug administration approved lenacapavir for the. Now, the well tolerated medication has been approved by the fda as a new preventative medication for all adults, as well as adolescents weighing at least 35kg, who are at risk of acquiring hiv. In the us, the food and drug administration (fda) has approved the novel lenacapavir – sold under the brand name yeztugo – a class of drugs known as capsid inhibitors, which provide almost 100% protection against hiv infection, which currently affects 1.3 million people every year.
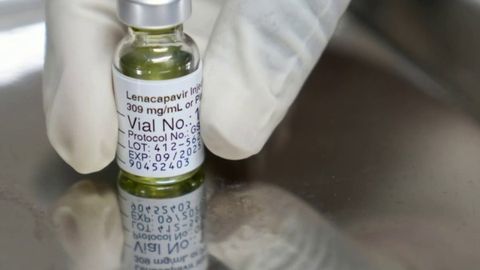
Fda Approves Powerful New Hiv Drug Haystack News Now, the well tolerated medication has been approved by the fda as a new preventative medication for all adults, as well as adolescents weighing at least 35kg, who are at risk of acquiring hiv. In the us, the food and drug administration (fda) has approved the novel lenacapavir – sold under the brand name yeztugo – a class of drugs known as capsid inhibitors, which provide almost 100% protection against hiv infection, which currently affects 1.3 million people every year. In 2022, gilead sciences, inc., received u.s. food and drug administration (fda) approval for lenacapavir, a twice yearly injectable medication for adults with multidrug resistant hiv. The u.s. food and drug administration (fda) approved a new, twice yearly shot — the first and only of its kind — to prevent hiv, the creator of the drug, gilead sciences, announced on. With a 99.9% success rate in preventing hiv transmission in clinical trials, this injectable therapy formally known as lenacapavir, offers a revolutionary alternative to daily pills and could dramatically reshape the global hiv landscape. This milestone marks the first preventive use of lenacapavir, previously approved for treatment of drug resistant hiv. the shift from daily pills to biannual injections could significantly improve adherence, especially in populations struggling with routine medication.

Nbc News Fda Approves First Injectable Hiv Prevention Drug Prep4all In 2022, gilead sciences, inc., received u.s. food and drug administration (fda) approval for lenacapavir, a twice yearly injectable medication for adults with multidrug resistant hiv. The u.s. food and drug administration (fda) approved a new, twice yearly shot — the first and only of its kind — to prevent hiv, the creator of the drug, gilead sciences, announced on. With a 99.9% success rate in preventing hiv transmission in clinical trials, this injectable therapy formally known as lenacapavir, offers a revolutionary alternative to daily pills and could dramatically reshape the global hiv landscape. This milestone marks the first preventive use of lenacapavir, previously approved for treatment of drug resistant hiv. the shift from daily pills to biannual injections could significantly improve adherence, especially in populations struggling with routine medication.

Fda Approves Long Lasting Hiv Prevention Drug Breitbart With a 99.9% success rate in preventing hiv transmission in clinical trials, this injectable therapy formally known as lenacapavir, offers a revolutionary alternative to daily pills and could dramatically reshape the global hiv landscape. This milestone marks the first preventive use of lenacapavir, previously approved for treatment of drug resistant hiv. the shift from daily pills to biannual injections could significantly improve adherence, especially in populations struggling with routine medication.

Fda Approves First Injectable Hiv Prevention Drug Mikey Live
Comments are closed.