Good Manufacturing Practices Gmps At Cdr

The Good Manufacturing Practices Gmps Contents This is a brief video of the good manufacturing practice (gmp) requirements at the center for dairy research. Good manufacturing practices (gmps) at cdr this is a brief video of the good manufacturing practice (gmp) requirements at the center for dairy research.

Good Manufacturing Practices Gmps In Feed Plant Pptx Agriculture Good manufacturing practices (gmp, also referred to as 'cgmp' or 'current good manufacturing practice') is the aspect of quality assurance that ensures that medicinal products are consistently produced and controlled to the quality standards appropriate to their intended use and as required by the product specification. In addition to the basic outreach efforts, the program can also provide assistance to your company in regards to grading, analyzing defects in cheese and butter as well as assistance in butter manufacturing practices. (1) all plant equipment and utensils used in manufacturing, processing, packing, or holding food must be so designed and of such material and workmanship as to be adequately cleanable, and must be adequately maintained to protect against allergen cross contact and contamination. The basic principles of good manufacturing practices help ensure product quality is consistent and safe to use. incorporating these will determine the scope and standards needed to pass gmp auditing.
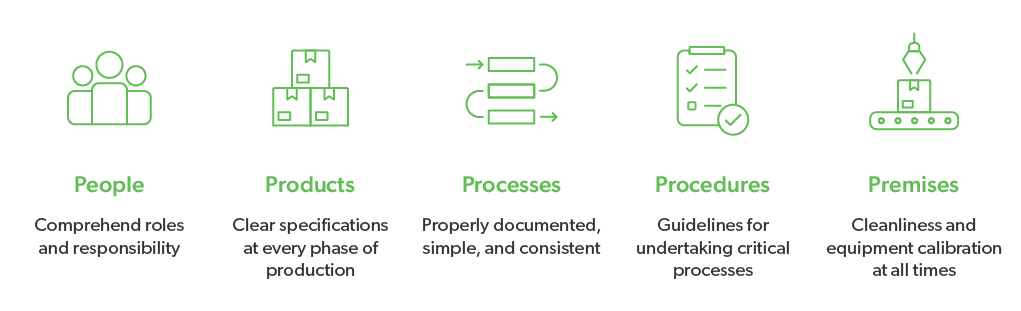
Ppt Good Manufacturing Practices Purpose And Principles Of 49 Off (1) all plant equipment and utensils used in manufacturing, processing, packing, or holding food must be so designed and of such material and workmanship as to be adequately cleanable, and must be adequately maintained to protect against allergen cross contact and contamination. The basic principles of good manufacturing practices help ensure product quality is consistent and safe to use. incorporating these will determine the scope and standards needed to pass gmp auditing. Guidelines of 19 march 2015 on the formalised risk assessment for ascertaining the appropriate good manufacturing practice for excipients of medicinal products for human use (all language versions are available here ). Learn about gmp guidelines, how to uphold gmp standards, and technology for commercial and industrial good manufacturing practices (gmps). Gmp compliance is the process of ensuring that food and pharmaceutical manufacturers produce and control products that meet good manufacturing practices, and minimize risks that they cannot eliminate through testing the final product. Good manufacturing practices (gmps) describe the methods, equipment, facilities, and controls for producing processed food. as the minimum sanitary and processing requirements for producing safe and wholesome food, they are an important part of regulatory control over the safety of the nation's food supply.
Comments are closed.