Free Radical Halogenation

Free Radical Halogenation Pdf Reactions of free radicals and radical ions. alkanes (the most basic of all organic compounds) undergo very few reactions. one of these reactions is halogenation, or the substitution of a single hydrogen on the alkane for a single halogen to form a haloalkane. Learn about the chemical reaction of halogenating alkanes and aromatics with uv light, involving free radical chain mechanism. find out the general mechanism, control, variations, and selectivity of this type of halogenation.
Free Radical Halogenation Alchetron The Free Social Encyclopedia In this tutorial, we are going to talk about the radical halogenation of alkanes. we’ll go over the intricacies of the mechanism, how to find the major products in this reaction, and discuss the most important points of each mechanistic step. here are a couple of simple examples of this reaction. These reactions include free radical halogenations of alkanes and free radical additions to alkenes. some aspects of these reactions cause them to be more complex than ionic reactions. Carbon free radicals are formed during halogenation of alkanes and orientation as well as reactivity depends on the type of free radical. it is known as free radical substitution reaction. Dive into the details of free radical halogenation, exploring its mechanisms, reaction conditions, and various applications in organic synthesis and beyond.

Ochem Free Radical Halogenation Flashcards Quizlet Carbon free radicals are formed during halogenation of alkanes and orientation as well as reactivity depends on the type of free radical. it is known as free radical substitution reaction. Dive into the details of free radical halogenation, exploring its mechanisms, reaction conditions, and various applications in organic synthesis and beyond. What is the mechanism of free radical halogenation? the mechanism of free radical halogenation involves three key steps: initiation, propagation, and termination. Free radical halogenation is a type of substitution reaction in organic chemistry where a hydrogen atom in an alkane is replaced by a halogen atom, such as chlorine or bromine, through a radical chain mechanism. this process is particularly important in the context of the properties of alkanes. The net reaction for halogenation seems straightforward, the mechanism is more complicated though, it go through multiple steps that include initiation, propagation and termination. In this article we will learn a simple guide on free halogenation of alkanes by general mechanism, free radical substitution and free radical halogenation.
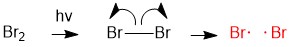
Free Radical Halogenation What is the mechanism of free radical halogenation? the mechanism of free radical halogenation involves three key steps: initiation, propagation, and termination. Free radical halogenation is a type of substitution reaction in organic chemistry where a hydrogen atom in an alkane is replaced by a halogen atom, such as chlorine or bromine, through a radical chain mechanism. this process is particularly important in the context of the properties of alkanes. The net reaction for halogenation seems straightforward, the mechanism is more complicated though, it go through multiple steps that include initiation, propagation and termination. In this article we will learn a simple guide on free halogenation of alkanes by general mechanism, free radical substitution and free radical halogenation.

Chemistry World Free Radical Halogenation A Question The net reaction for halogenation seems straightforward, the mechanism is more complicated though, it go through multiple steps that include initiation, propagation and termination. In this article we will learn a simple guide on free halogenation of alkanes by general mechanism, free radical substitution and free radical halogenation.
Comments are closed.