Forms Of Carbon

Amorphous Forms Of Carbon Physics Wallah 55 Off Learn about the different forms of carbon, such as diamond, graphite, fullerenes, and nanotubes. explore their properties, structures, and applications in various fields. Different forms, or allotropes, of carbon are diamond, graphite, fullerene, coal, and charcoal. in a diamond, each carbon atom is bonded to four other carbon atoms forming a rigid structure that makes the diamond very hard.

Carbon And Its Various Forms Carbon Carbon Carbon Is A Non Metallic El Elemental carbon exists in several forms, each of which has its own physical characteristics. two of its well defined forms, diamond and graphite, are crystalline in structure, but they differ in physical properties because the arrangements of the atoms in their structures are dissimilar. Learn about the different forms and properties of carbon, such as diamond, graphite, and carbides. explore the chemical reactions and compounds of carbon, including co2, carbonates, and fullerenes. Carbon (pronounced as kar ben) is a soft nonmetal denoted by the chemical symbol c. it forms several allotropes including diamond, graphite, graphene, and fullerene. Carbon with its both ordered and disordered configuration covers a wide variety of materials and properties ranging from graphite, a black, soft, and electrically conductive species to diamond, a glittery, extremely hard, and near perfect electrical insulator.

Carbon Less Forms Efficient Printing Solutions Laguna Beach Carbon (pronounced as kar ben) is a soft nonmetal denoted by the chemical symbol c. it forms several allotropes including diamond, graphite, graphene, and fullerene. Carbon with its both ordered and disordered configuration covers a wide variety of materials and properties ranging from graphite, a black, soft, and electrically conductive species to diamond, a glittery, extremely hard, and near perfect electrical insulator. Carbon is capable of forming many allotropes in addition to the well known diamond and graphite forms. the physical properties of carbon vary widely with the allotropic form. for example, diamond is highly transparent, but graphite is opaque and black. Carbon is a remarkable element, forming the chemical backbone of all known life and serving as a foundational material in modern engineering. this versatility stems from its unique ability to bond with itself in various configurations, creating different physical forms known as allotropes. Learn about carbon, an extraordinary element that occurs in more different forms than any other element in the periodic table. discover its physical and chemical properties, its allotropes (such as diamond and graphite), and its role in organic chemistry and living organisms. Well known allotropes include graphite, diamond, amorphous carbon, and fullerenes. the physical properties of carbon vary widely with the allotropic form. for example, graphite is opaque and black, while diamond is highly transparent.

Superhard Forms Of Carbon Expected To Rival Diamonds Futurity Carbon is capable of forming many allotropes in addition to the well known diamond and graphite forms. the physical properties of carbon vary widely with the allotropic form. for example, diamond is highly transparent, but graphite is opaque and black. Carbon is a remarkable element, forming the chemical backbone of all known life and serving as a foundational material in modern engineering. this versatility stems from its unique ability to bond with itself in various configurations, creating different physical forms known as allotropes. Learn about carbon, an extraordinary element that occurs in more different forms than any other element in the periodic table. discover its physical and chemical properties, its allotropes (such as diamond and graphite), and its role in organic chemistry and living organisms. Well known allotropes include graphite, diamond, amorphous carbon, and fullerenes. the physical properties of carbon vary widely with the allotropic form. for example, graphite is opaque and black, while diamond is highly transparent.
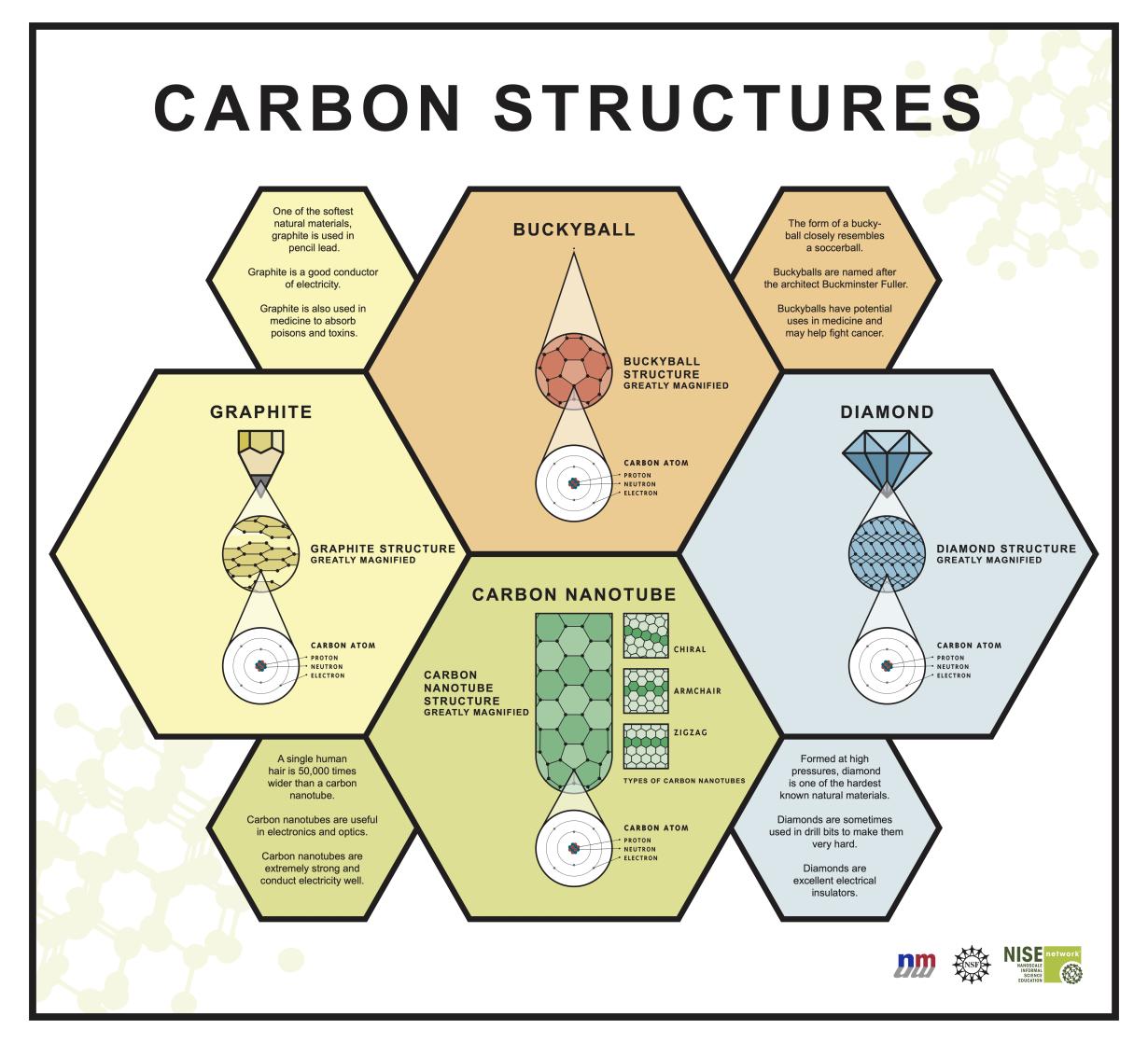
Carbon Nise Network Learn about carbon, an extraordinary element that occurs in more different forms than any other element in the periodic table. discover its physical and chemical properties, its allotropes (such as diamond and graphite), and its role in organic chemistry and living organisms. Well known allotropes include graphite, diamond, amorphous carbon, and fullerenes. the physical properties of carbon vary widely with the allotropic form. for example, graphite is opaque and black, while diamond is highly transparent.
Comments are closed.