Finding The Minimum Required Reagent Concentration For Selective Precipitation Practice Problems

Practice Problems Precipitation Titration Pdf Molar Concentration To find the concentration needed to start precipitation, write the ionic solid dissociation, express \ (k {sp}\) in terms of ion concentrations, and determine the minimum reagent concentration that makes the system just reach the precipitation threshold. Example #3: a solution of 0.10 m (each) ni 2 and cu 2 are separated using selective precipitation by the addition of solid na 2 co 3. assuming no volume change upon this addition, how much of the first precipitated ion (in %) remains at the point where the second ion begins to precipitate?.

Selective Precipitation Worksheet Chemistry Problems In this video i go over a couple of practice problems on how to find the minimum required reagent concentration that is needed to selectively precipitate one ion over another .more. This document contains 11 practice problems about solution stoichiometry and selective precipitation for a chemistry 11 class. Knowledge of ksp values will allow you to be able to predict whether or not a precipitate will form when two solutions are mixed together. for example, suppose that a known solution of barium chloride is mixed with a known solution of sodium sulfate. Write an equation for the reaction of solid silicon dioxide with hydrofluoric acid to yield gaseous silicon tetrafluoride and liquid water. the mineral fluorite (calcium fluoride) occurs extensively in illinois.

Finding The Limiting Reagent In A Precipitation Reaction Chemistry Knowledge of ksp values will allow you to be able to predict whether or not a precipitate will form when two solutions are mixed together. for example, suppose that a known solution of barium chloride is mixed with a known solution of sodium sulfate. Write an equation for the reaction of solid silicon dioxide with hydrofluoric acid to yield gaseous silicon tetrafluoride and liquid water. the mineral fluorite (calcium fluoride) occurs extensively in illinois. Selective precipitation is used to obtain a solid out of solution. we can calculate which substance will precipitate in a reaction using solubilities of different substances. By increasing the counter ion concentration in a controlled manner, ions in solution may be precipitated individually, assuming their compound solubilities are adequately different. Practice problem: selective precipitation example 17.13 finding the minimum required reagent concentration for selective precipitation the magnesium and calcium ions present in seawater ( [mg] 0.059 m and (ca?') 0,011 m) can be separated by selective precipitation with koh. By increasing the counter ion concentration in a controlled manner, ions in solution may be precipitated individually, assuming their compound solubilities are adequately different.
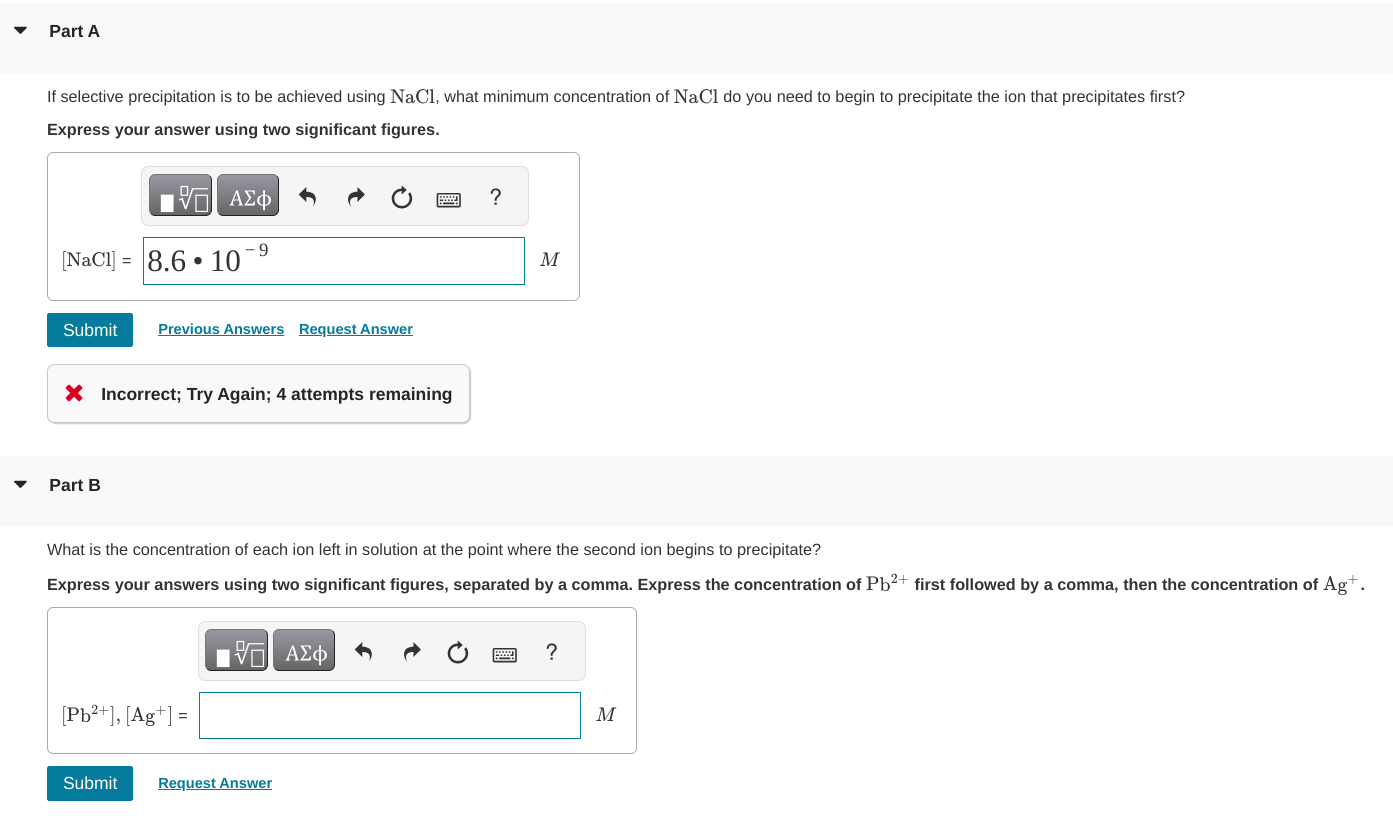
If Selective Precipitation Is To Be Achieved Using Chegg Selective precipitation is used to obtain a solid out of solution. we can calculate which substance will precipitate in a reaction using solubilities of different substances. By increasing the counter ion concentration in a controlled manner, ions in solution may be precipitated individually, assuming their compound solubilities are adequately different. Practice problem: selective precipitation example 17.13 finding the minimum required reagent concentration for selective precipitation the magnesium and calcium ions present in seawater ( [mg] 0.059 m and (ca?') 0,011 m) can be separated by selective precipitation with koh. By increasing the counter ion concentration in a controlled manner, ions in solution may be precipitated individually, assuming their compound solubilities are adequately different.
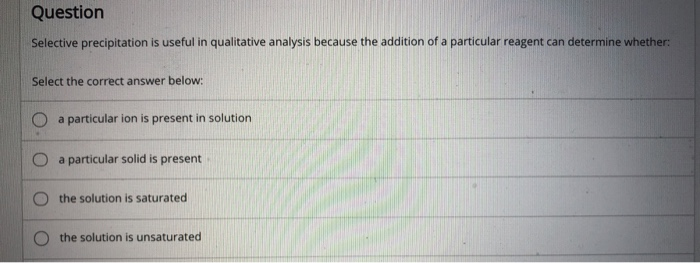
Solved Question Selective Precipitation Is Useful In Chegg Practice problem: selective precipitation example 17.13 finding the minimum required reagent concentration for selective precipitation the magnesium and calcium ions present in seawater ( [mg] 0.059 m and (ca?') 0,011 m) can be separated by selective precipitation with koh. By increasing the counter ion concentration in a controlled manner, ions in solution may be precipitated individually, assuming their compound solubilities are adequately different.

Selective Precipitation Calculations Pdf Solubility Precipitation
Comments are closed.