Figure 3 From Metamorphic Proteins Under A Computational Microscope

Pdf Metamorphic Proteins Under A Computational Microscope Lessons These efforts in studying the refolding landscapes of two quintessential metamorphic proteins, rfah and kaib, are presented, using simplified dual basin structure based models (sbms), rigorously footed on the energy landscape theory of protein folding and the principle of minimal frustration. The dramatic nature of rfah structural transformation and the richness of its evolutionary history makes for an excellent model for studying how metamorphic proteins switch folds.

Images Of Sample Metamorphic Rock Under A Multi Functional Microscope In this review, we describe the current advances in biophysically and functionally ascertaining the structural interconversions of metamorphic proteins and how coevolution can be harnessed to identify novel metamorphic proteins from sequence information. Metamorphic proteins constitute unexpected paradigms of the protein folding problem, as their sequences encode two alternative folds, which reversibly interconvert within biologically relevant timescales to trigger different cellular responses. Proteins that are able to switch between different folds correlate their structural and functional changes, however, crowding effects on fold switching proteins are not fully understood. Comparison of our computational results with the available experimental data show that the md based thermodynamics can explain the experimentally observed variation of the conformational equilibrium between the two proteins.
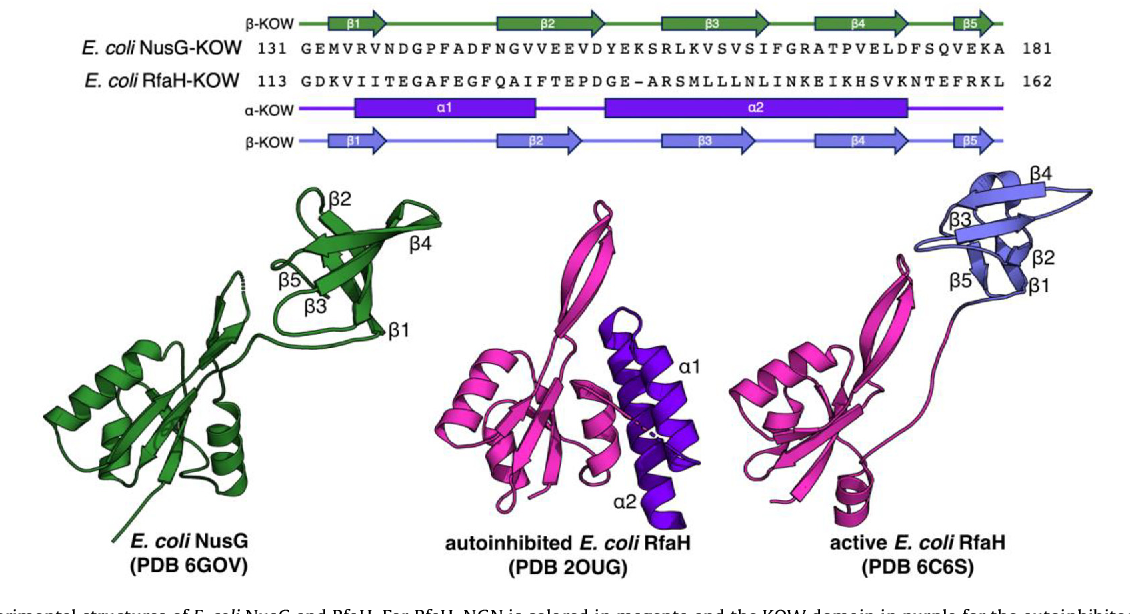
Figure 3 From Metamorphic Proteins Under A Computational Microscope Proteins that are able to switch between different folds correlate their structural and functional changes, however, crowding effects on fold switching proteins are not fully understood. Comparison of our computational results with the available experimental data show that the md based thermodynamics can explain the experimentally observed variation of the conformational equilibrium between the two proteins. Here, we discuss noncanonical protein folding patterns, with an emphasis on metamorphic proteins, and we review known metamorphic proteins that occur naturally and that have been engineered in the laboratory. A small but growing number of “metamorphic” proteins adopt different folded conformations for the same amino acid sequence in native conditions. unlike prions, they undergo reversible conformational chan ges.

Representative Examples Of Metamorphic Proteins With 3 Dimensional Here, we discuss noncanonical protein folding patterns, with an emphasis on metamorphic proteins, and we review known metamorphic proteins that occur naturally and that have been engineered in the laboratory. A small but growing number of “metamorphic” proteins adopt different folded conformations for the same amino acid sequence in native conditions. unlike prions, they undergo reversible conformational chan ges.

Representative Examples Of Metamorphic Proteins With 3 Dimensional
Comments are closed.