Excess Reagent

Limiting And Excess Reagent Worksheet Pdf Learn what an excess reagent is and how to identify it in a chemical reaction. see examples of excess reagent and limiting reagent with balanced equations and calculations. The reactant that is entirely consumed is called the limiting reactant; t he other reactant or reactants are present in excess. a crucial skill in evaluating the conditions of a chemical process is to determine which reactant is the limiting reactant and which is are the excess reactant (s).

Excess Reagent Definition And Examples Once the limiting reactant gets used up, the reaction has to stop and cannot continue and there is extra of the other reactants left over. those are called the excess reactants. Learn how to determine the excess reactant in a chemical reaction with step by step explanations. understand its role in stoichiometry, how to calculate leftover amounts, and practice with real world examples. Learn how to calculate the amount of excess reactant left over after a chemical reaction using the stoichiometry of the limiting reactant. follow the steps and examples for mno2 and hcl reaction. Master the process of determining the amount of reactant left over when the limiting reactant is consumed. follow clear steps and see worked examples to deepen your stoichiometry understanding.
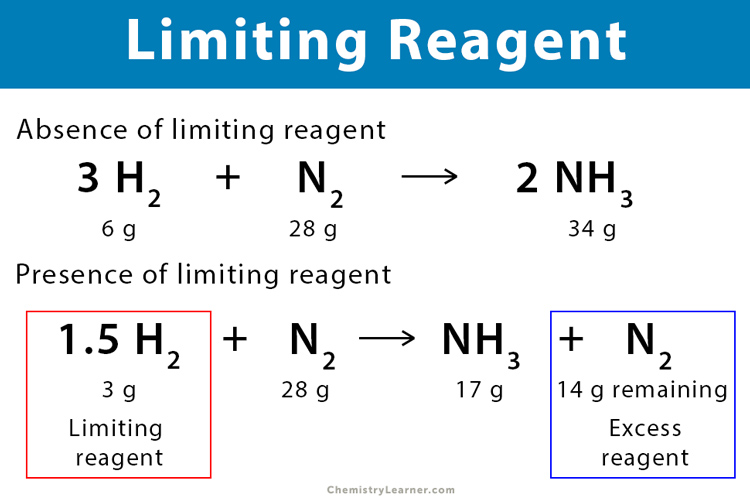
Excess Reagent Definition And Examples Learn how to calculate the amount of excess reactant left over after a chemical reaction using the stoichiometry of the limiting reactant. follow the steps and examples for mno2 and hcl reaction. Master the process of determining the amount of reactant left over when the limiting reactant is consumed. follow clear steps and see worked examples to deepen your stoichiometry understanding. Learn how to find the limiting and excess reactant in chemistry with our easy to follow guide. try our problems and check your understanding!. Learn how to identify limiting and excess reactants with our step by step stoichiometry guide. calculate theoretical yield easily. master your chemistry exams now!. While the limiting reagent dictates the maximum yield of products formed, the excess reagent is the leftover reactant that remains after the reaction has concluded. What is excess reagent? during a chemical reaction, one of the reactants is found to be present in a higher quantity than what is necessary for the completion of the reaction.

Excess Reagent Definition And Examples Learn how to find the limiting and excess reactant in chemistry with our easy to follow guide. try our problems and check your understanding!. Learn how to identify limiting and excess reactants with our step by step stoichiometry guide. calculate theoretical yield easily. master your chemistry exams now!. While the limiting reagent dictates the maximum yield of products formed, the excess reagent is the leftover reactant that remains after the reaction has concluded. What is excess reagent? during a chemical reaction, one of the reactants is found to be present in a higher quantity than what is necessary for the completion of the reaction.

Excess Reagent Definition And Examples While the limiting reagent dictates the maximum yield of products formed, the excess reagent is the leftover reactant that remains after the reaction has concluded. What is excess reagent? during a chemical reaction, one of the reactants is found to be present in a higher quantity than what is necessary for the completion of the reaction.
Comments are closed.