Ethanoic Acid Ethanol Ester Reaction

Reaction Of Ethanol With Ethanoic Acid Reaction Of An Ester With A Base Learn about the formation of an ester for your igcse chemistry exam. also find information of the formation of ethanoic acid. learn more. To make a small ester like ethyl ethanoate, you can gently heat a mixture of ethanoic acid and ethanol in the presence of concentrated sulfuric acid, and distil off the ester as soon as it is formed.
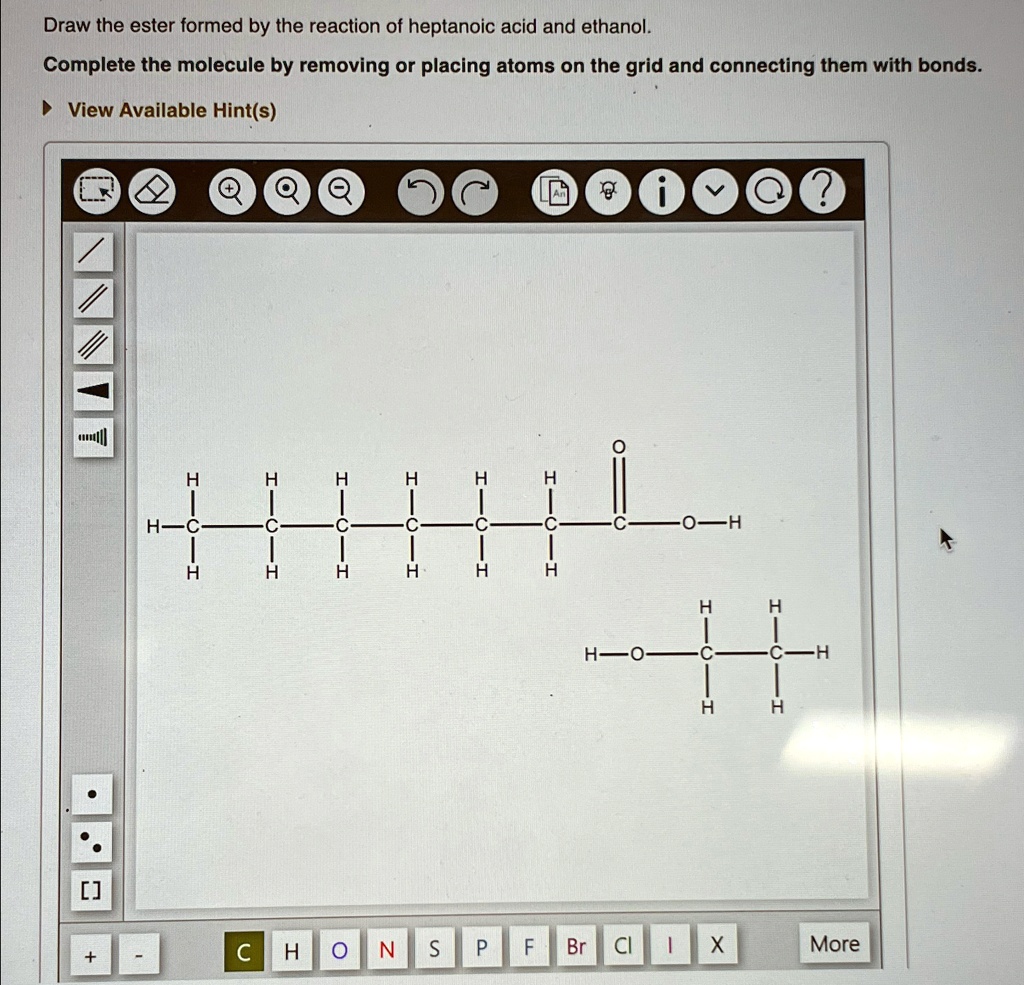
Draw The Ester Formed By The Reaction Of Heptanoic Acid And Ethanol It uses the formation of ethyl ethanoate from ethanoic acid and ethanol as a typical example. ethanoic acid reacts with ethanol in the presence of concentrated sulphuric acid as a catalyst to produce the ester, ethyl ethanoate. the reaction is slow and reversible. Investigate the reactions between a range of alcohols and acids by producing a variety of esters in this class experiment. includes kit list and safety instructions. The document describes a chemistry laboratory experiment on the esterification reaction between ethanol and ethanoic acid using concentrated sulfuric acid to produce the ester ethyl ethanoate. How does ethanoic acid react with alcohols? ethanoic acid reacts with alcohols in the presence of a catalyst to form an ester and water. in more detail, this reaction is known as esterification. when ethanoic acid (ch3cooh) reacts with an alcohol (r oh), an ester (r cooh) and water (h2o) are formed.
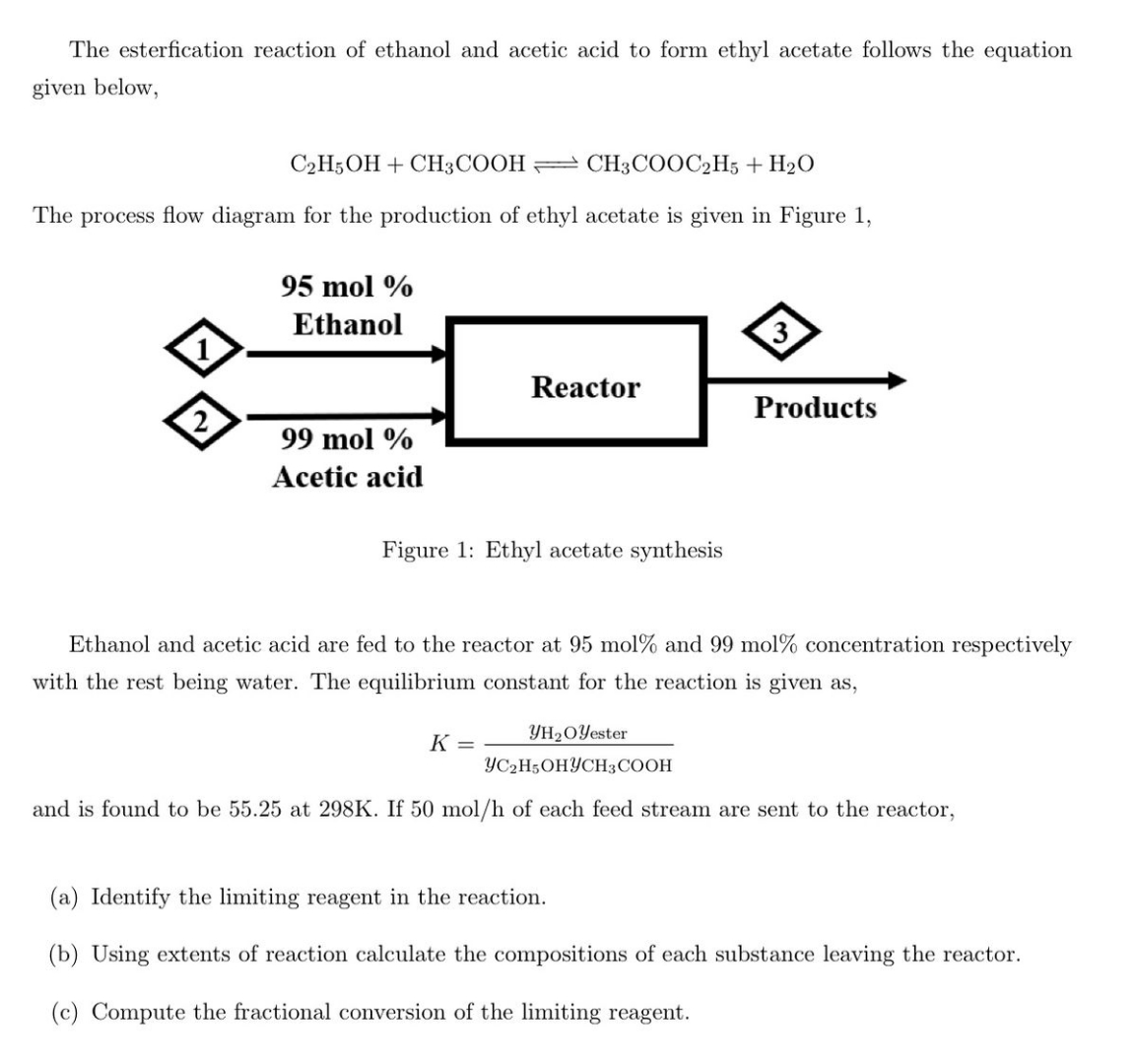
Solved The Esterfication Reaction Of Ethanol And Acetic Acid Chegg The document describes a chemistry laboratory experiment on the esterification reaction between ethanol and ethanoic acid using concentrated sulfuric acid to produce the ester ethyl ethanoate. How does ethanoic acid react with alcohols? ethanoic acid reacts with alcohols in the presence of a catalyst to form an ester and water. in more detail, this reaction is known as esterification. when ethanoic acid (ch3cooh) reacts with an alcohol (r oh), an ester (r cooh) and water (h2o) are formed. To form an ester from ethanoic acid and ethanol, we will perform an esterification reaction. this reaction involves the condensation of an alcohol and a carboxylic acid, resulting in the formation of an ester and water. Ans. ethanoic acid can be formed through the esterification reaction between ethanol (alcohol) and acetic acid (carboxylic acid), producing ethyl acetate (ester) and water. Ethanoic acid and ethanol will react together to form an ester, ethyl ethanoate. the reaction is a condensation because water is made, but is also known as esterification because an. The reaction between ethanoic acid and absolute ethanol to produce an ester is known as esterification. in this reaction, ethanoic acid (ch3cooh) reacts with ethanol (c2h5oh) in the presence of an acid catalyst, typically sulfuric acid (h2so4).
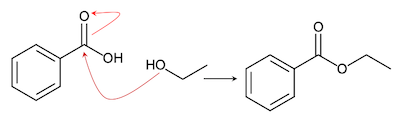
Draw The Ester That Is Formed From The Reaction Of Benzoic Acid And To form an ester from ethanoic acid and ethanol, we will perform an esterification reaction. this reaction involves the condensation of an alcohol and a carboxylic acid, resulting in the formation of an ester and water. Ans. ethanoic acid can be formed through the esterification reaction between ethanol (alcohol) and acetic acid (carboxylic acid), producing ethyl acetate (ester) and water. Ethanoic acid and ethanol will react together to form an ester, ethyl ethanoate. the reaction is a condensation because water is made, but is also known as esterification because an. The reaction between ethanoic acid and absolute ethanol to produce an ester is known as esterification. in this reaction, ethanoic acid (ch3cooh) reacts with ethanol (c2h5oh) in the presence of an acid catalyst, typically sulfuric acid (h2so4).
Solved Acetic Acid And Ethanol React To Form An Ester Product As Ethanoic acid and ethanol will react together to form an ester, ethyl ethanoate. the reaction is a condensation because water is made, but is also known as esterification because an. The reaction between ethanoic acid and absolute ethanol to produce an ester is known as esterification. in this reaction, ethanoic acid (ch3cooh) reacts with ethanol (c2h5oh) in the presence of an acid catalyst, typically sulfuric acid (h2so4).
Comments are closed.