Ester Functional Group Chemistry Steps
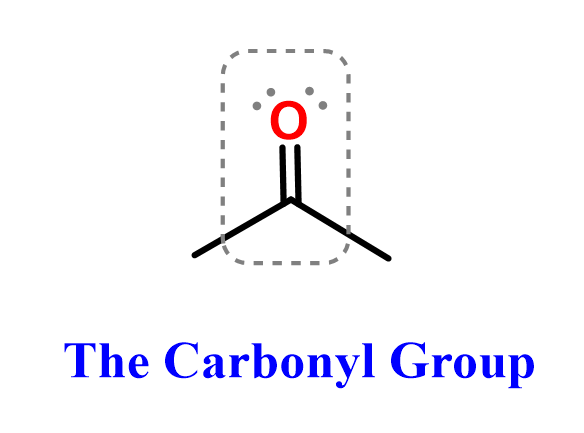
Ester Functional Group Chemistry Steps Esters are widely found in fragrances, flavorings, fats, oils, and biological molecules, and they play key roles in condensation and hydrolysis reactions. since esters contain a carbonyl group, the c=o bond remains the central feature of their reactivity. By recognizing that the steps in the acidic hydrolysis of an ester are exactly the same as those in a fischer esterification (but in the reverse order!), you can again minimize the amount of memorization that you must undertake.
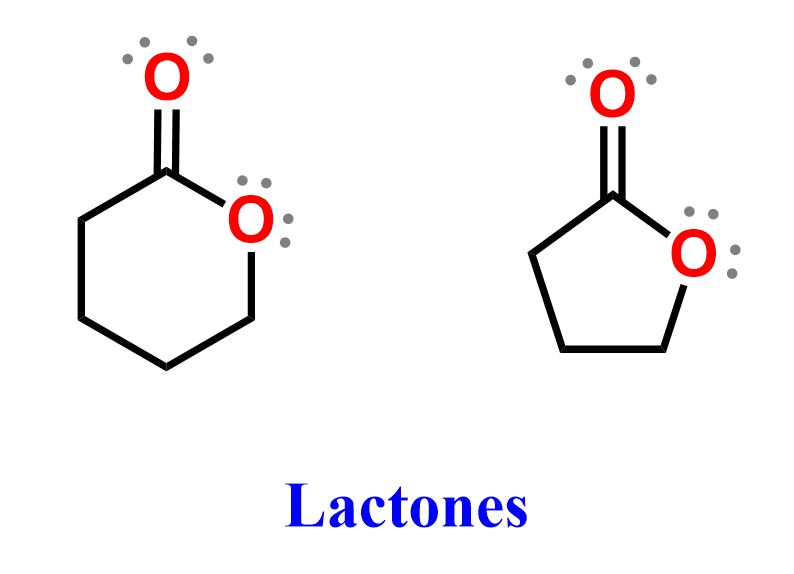
Ester Functional Group Chemistry Steps In this tutorial you will learn about the basic properties and structure of an ester functional group. you will also learn about esterification and its mechanism. Understand the process of making and naming esters in igcse chemistry and their significance in food flavorings and perfumes, along with examples. Esters can be made using a carboxylic acid and alcohol, in a process called esterification. a carboxylic acid and an alcohol are mixed in the presence of an acid catalyst (usually concentrated sulfuric acid). reflux conditions are required as energy is needed to start the reaction. Step 6: number the parent hydrocarbon chain from the end that produces the lowest set of locants for, in order of precedence, functional groups, double and triple bonds and side chains.

Ester Functional Group Chemistry Steps Esters can be made using a carboxylic acid and alcohol, in a process called esterification. a carboxylic acid and an alcohol are mixed in the presence of an acid catalyst (usually concentrated sulfuric acid). reflux conditions are required as energy is needed to start the reaction. Step 6: number the parent hydrocarbon chain from the end that produces the lowest set of locants for, in order of precedence, functional groups, double and triple bonds and side chains. Find out why fats are solids while oils are liquids by revising esters for higher chemistry. learn how the important reactions involved work. There are several ways to prepare esters, and in this lesson, we’ll go over the most common ones you’ll see in organic chemistry. the most famous is the fischer esterification, where a carboxylic acid is reacted with an alcohol under acidic conditions and elevated temperature. Esters are organic compounds that contain a functional group composed of a carbonyl group (c=o) bonded to an oxygen atom (o), which is further bonded to an alkyl or aryl group (r). the general structure of an ester is rcoor', where r and r' represent alkyl or aryl groups. Esters are formed through reactions between an acid and an alcohol with the elimination of water. an example of this is the reaction of acetic acid with an alcohol, which yields an acetic ester and water.
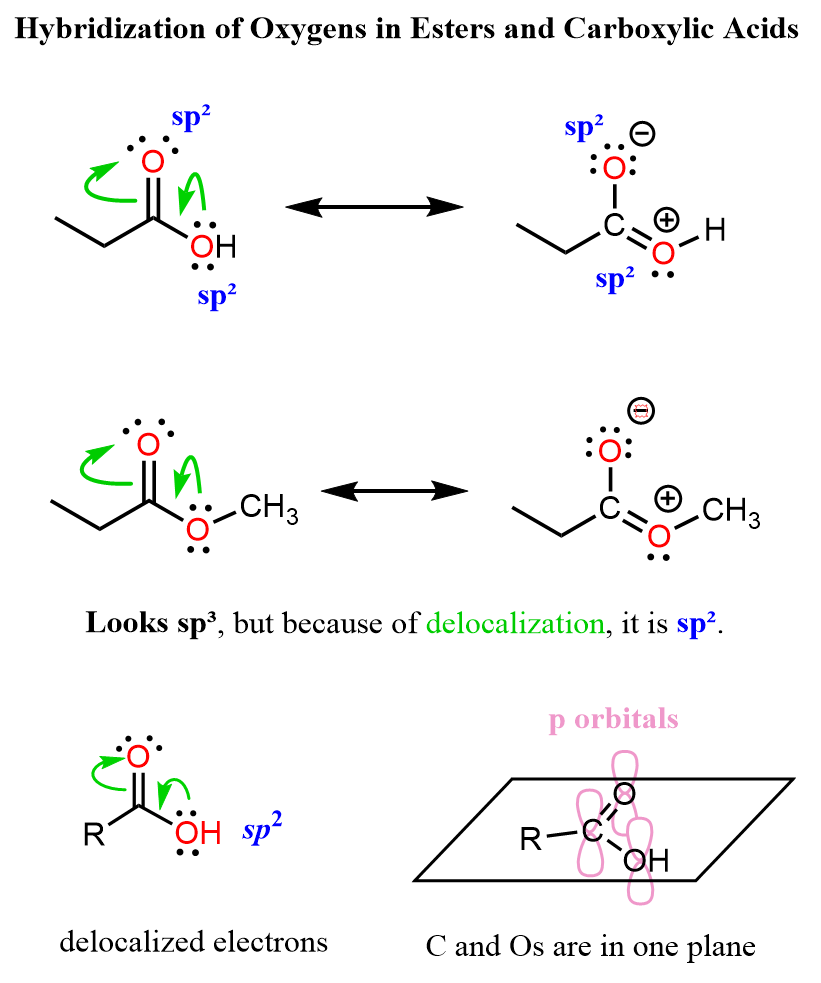
Ester Functional Group Chemistry Steps Find out why fats are solids while oils are liquids by revising esters for higher chemistry. learn how the important reactions involved work. There are several ways to prepare esters, and in this lesson, we’ll go over the most common ones you’ll see in organic chemistry. the most famous is the fischer esterification, where a carboxylic acid is reacted with an alcohol under acidic conditions and elevated temperature. Esters are organic compounds that contain a functional group composed of a carbonyl group (c=o) bonded to an oxygen atom (o), which is further bonded to an alkyl or aryl group (r). the general structure of an ester is rcoor', where r and r' represent alkyl or aryl groups. Esters are formed through reactions between an acid and an alcohol with the elimination of water. an example of this is the reaction of acetic acid with an alcohol, which yields an acetic ester and water.

Ester Functional Group Chemistry Steps Esters are organic compounds that contain a functional group composed of a carbonyl group (c=o) bonded to an oxygen atom (o), which is further bonded to an alkyl or aryl group (r). the general structure of an ester is rcoor', where r and r' represent alkyl or aryl groups. Esters are formed through reactions between an acid and an alcohol with the elimination of water. an example of this is the reaction of acetic acid with an alcohol, which yields an acetic ester and water.

Ester Functional Group Chemistry Steps
Comments are closed.