Error Analysis Example Pdf Titration Chemistry

Chemistry Lab Report Titration Pdf Titration Chemistry This document discusses the process of determining important errors in a titration experiment. it distinguishes between random errors, which cancel out with repetition, and systematic errors, which consistently make results too high or too low. The purpose of the error analysis section of the lab report is to determine the most important errors and the effect that those errors have on the final result.

Titration Pdf Titration Chemistry Many errors in analytical analysis arise from poor sample preparation or instrument set up. this chapter will guide you through common preparation errors for both potentiometric and karl fischer titration methods and provide suggestions on how to avoid them. Error analysis example in titration. this document has been uploaded by a student, just like you, who decided to remain anonymous. experimental error is one of the important skills t hat we need to learn to be effective scientists. in. the corresponding text that would be written in a l ab report for ch141. In this experiment, the analysis involves 2 neutralisation reactions: between a solution of a weak acid (acetic acid ch3cooh) and a solution of a strong base (naoh). an acid base titration is complete when exactly the right amount of base has been added to react with all of the acid in the solution. The aim of this booklet is to explore titration from a historical, theoretical and practical point of view, dealing first with preset end point titration then with inflection point titration.

Error Analysis Example In Titration Chem 3300 Studocu In this experiment, the analysis involves 2 neutralisation reactions: between a solution of a weak acid (acetic acid ch3cooh) and a solution of a strong base (naoh). an acid base titration is complete when exactly the right amount of base has been added to react with all of the acid in the solution. The aim of this booklet is to explore titration from a historical, theoretical and practical point of view, dealing first with preset end point titration then with inflection point titration. Before beginning an analysis ask, “what maximum error can be tolerated in the result?” the answer to this question often determines the method chosen and the time required to complete the analysis. Equivalence point is the point in the titration where the acetic acid in the conical flask and the naoh delivered from the burette have reacted completely according to the stoichiometric ratio of the balanced chemical equation for the reaction. Suppose you have a new analytical method for measuring %ni in a metal sample. you make four measurements of the nickel concentration and get the following results:. Titration methods are based on determining the quantity of a reagent of known concentration that is required to react completely with the analyte. the reagent may be a standard solution of a chemical or an electric current of known magnitude.
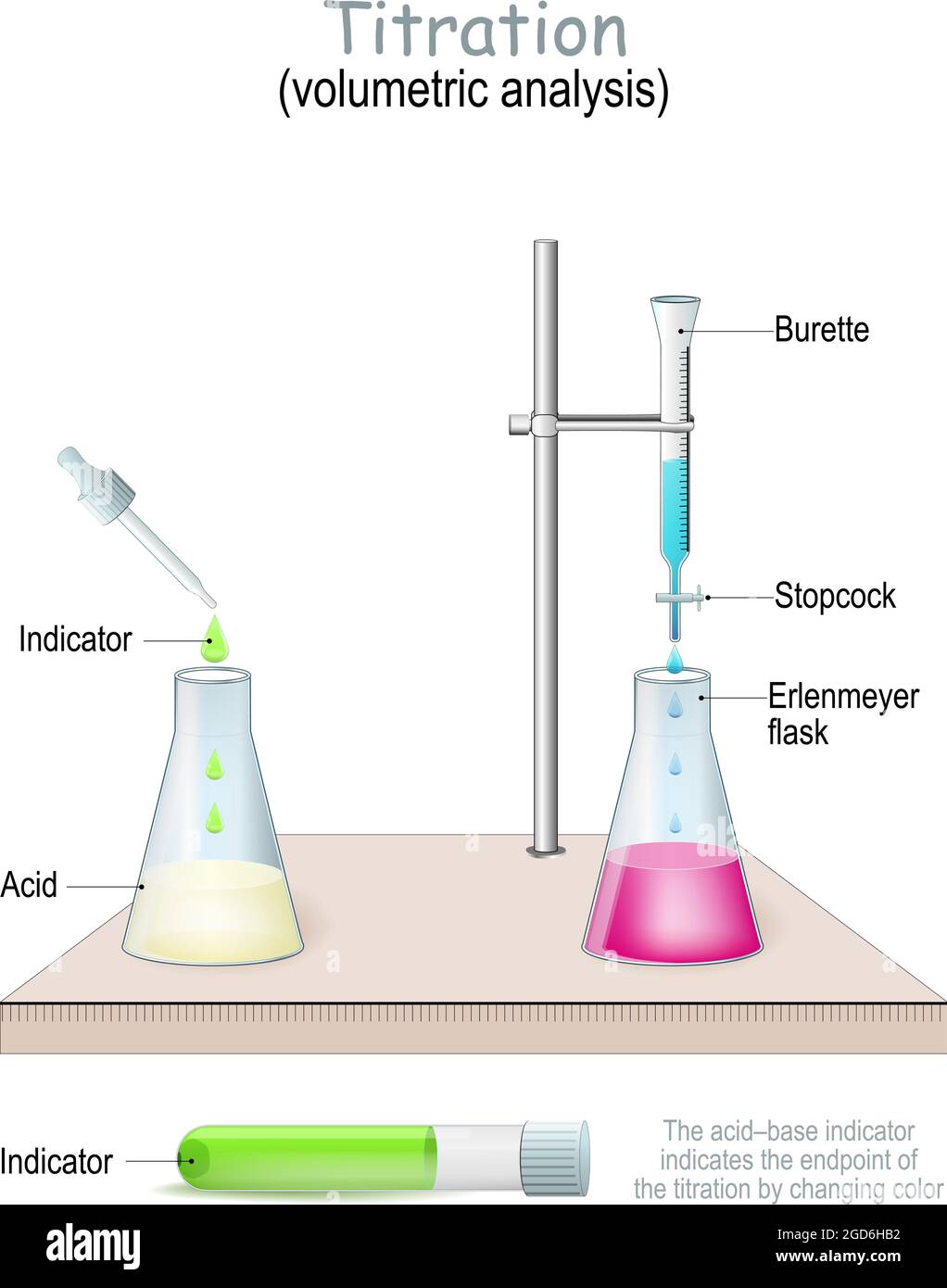
Titration Before beginning an analysis ask, “what maximum error can be tolerated in the result?” the answer to this question often determines the method chosen and the time required to complete the analysis. Equivalence point is the point in the titration where the acetic acid in the conical flask and the naoh delivered from the burette have reacted completely according to the stoichiometric ratio of the balanced chemical equation for the reaction. Suppose you have a new analytical method for measuring %ni in a metal sample. you make four measurements of the nickel concentration and get the following results:. Titration methods are based on determining the quantity of a reagent of known concentration that is required to react completely with the analyte. the reagent may be a standard solution of a chemical or an electric current of known magnitude.

Error Analysis Example Pdf Titration Chemistry Suppose you have a new analytical method for measuring %ni in a metal sample. you make four measurements of the nickel concentration and get the following results:. Titration methods are based on determining the quantity of a reagent of known concentration that is required to react completely with the analyte. the reagent may be a standard solution of a chemical or an electric current of known magnitude.
Comments are closed.