Emulsions To Class Pdf Emulsion Chemical Polarity

Emulsions To Class Pdf Emulsion Chemical Polarity They can be categorized into oil in water, water in oil, microemulsions, and multiple emulsions, each with distinct properties and applications. the document also discusses the theories of emulsification, types of emulsifying agents, and the advantages and disadvantages of microemulsions. Several classes may be distinguished: oil in water (o w), water in oil (w o), and oil in oil (o o). the latter class may be exemplified by an emulsion consisting of a polar oil (e.g., propylene glycol) dispersed in a nonpolar oil (paraffinic oil) and vice versa.

Emulsion Emulsifying Agent Types Properties Examples The purpose of this review article is to provide information about types of emulsions, stability mechanisms and rheological studies as well as factor affecting the stability of emulsions. Emulsions: introduction, classification, and formulation overview emulsions are biphasic liquid systems consisting of two immiscible liquids, where one liquid is dispersed as droplets within another using an emulsifying agent. Emulsions can be classified according to different criteria. in the classic type of emulsion or broad sense, the two immiscible liquids involved are water and oil. Definition: an emulsion is a heterogeneous mixture of two immiscible liquids where one liquid (the dispersed phase) is dispersed in the form of small droplets throughout the other liquid (the continuous phase).

Emulsion And Emulsifiers Pdf Emulsions can be classified according to different criteria. in the classic type of emulsion or broad sense, the two immiscible liquids involved are water and oil. Definition: an emulsion is a heterogeneous mixture of two immiscible liquids where one liquid (the dispersed phase) is dispersed in the form of small droplets throughout the other liquid (the continuous phase). An emulsion is made up of more than just water and oil; it can also include solid particles and even gas. due to the unfavourable interaction between the oil and water phases, an emulsion is inherently an unstable system. The hydrophilic lipophilic balance (hlb) of emulsifiers determines emulsion types, with low hlb values favoring water in oil emulsions and high values favoring oil in water emulsions. Emulsions do not form without mechanical agitation strong interfacial repulsion and strong insolubility of liquids allow droplets to remain dispersed over many years. The type of emulsifier and or surfactant employed is one of the most important factors to consider when developing an emulsion. emulsifiers and surfactants are typically amphiphilic molecules, which means they have both polar and nonpolar regions.
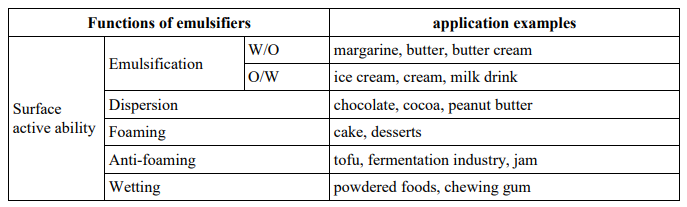
Emulsions Properties Types And Emulsifying Agents Food Additives An emulsion is made up of more than just water and oil; it can also include solid particles and even gas. due to the unfavourable interaction between the oil and water phases, an emulsion is inherently an unstable system. The hydrophilic lipophilic balance (hlb) of emulsifiers determines emulsion types, with low hlb values favoring water in oil emulsions and high values favoring oil in water emulsions. Emulsions do not form without mechanical agitation strong interfacial repulsion and strong insolubility of liquids allow droplets to remain dispersed over many years. The type of emulsifier and or surfactant employed is one of the most important factors to consider when developing an emulsion. emulsifiers and surfactants are typically amphiphilic molecules, which means they have both polar and nonpolar regions.

Chapstick Engineering Step 2 Pdf Chemical Polarity Emulsion Emulsions do not form without mechanical agitation strong interfacial repulsion and strong insolubility of liquids allow droplets to remain dispersed over many years. The type of emulsifier and or surfactant employed is one of the most important factors to consider when developing an emulsion. emulsifiers and surfactants are typically amphiphilic molecules, which means they have both polar and nonpolar regions.
Comments are closed.