Electrostatic Attraction

Electrostatic Attraction Electrostatic phenomena arise from the forces that electric charges exert on each other. such forces are described by coulomb's law. Electrostatic attraction is a fundamental force that acts between particles carrying an electric charge. this phenomenon is one of the four basic forces of nature, responsible for almost every interaction we observe in daily life, from subatomic bonds to large scale industrial processes.

Electrostatic Attraction The charge and size of a ligand atom and or metal ion can effect the electrostatic attraction between the two, in turn effecting the attractive force between metal and ligand. this comes from coulomb's law. in general, the higher the charge, the stronger the attraction between metal and ligand. Learn about the electrostatic force, the attraction or repulsion between two charged particles, and its formula based on coulomb's law. see examples of electrostatic force in daily life and its applications in physics. Explore the science behind electrostatic attraction—how it influences atomic interactions, its role in everyday life, and applications in chemistry. Surprisingly, the answer is yes, and the force is attractive. the reason is that, under the influence of the electric field of a charged object, the negatively charged electrons and the positively charged nuclei within the atoms and molecules are subjected to forces in opposite directions.
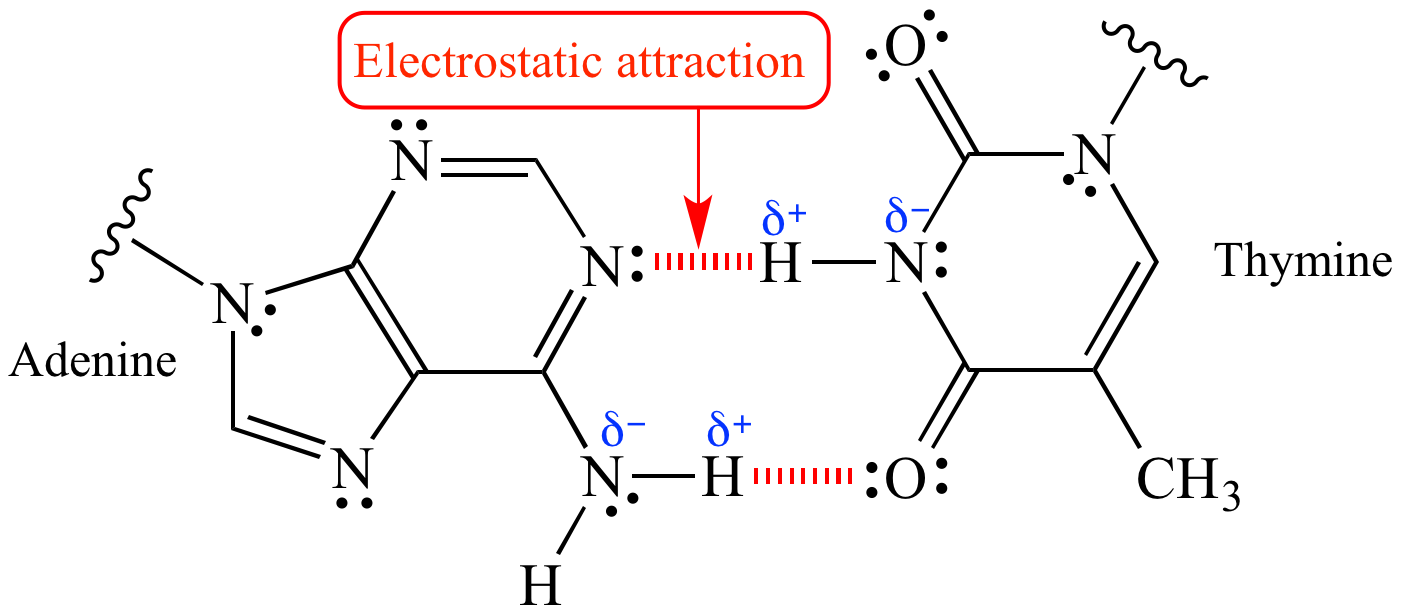
Electrostatic Attraction Explore the science behind electrostatic attraction—how it influences atomic interactions, its role in everyday life, and applications in chemistry. Surprisingly, the answer is yes, and the force is attractive. the reason is that, under the influence of the electric field of a charged object, the negatively charged electrons and the positively charged nuclei within the atoms and molecules are subjected to forces in opposite directions. Electrostatic attraction is the force of attraction between oppositely charged particles or objects. it is a fundamental concept in the study of ionic bonding, where the attractive force between positively and negatively charged ions holds the ionic compound together. The second big difference between the electrostatic force and gravity is that electrostatic forces come in two types: attractive and repulsive. there are two types of charge, and that means the force can go in one of two directions: toward and away. meanwhile, gravity is only an attractive force. Explore coulomb's law, which describes the electrostatic forces two electric charges exert on each other. learn how the strengths of the electrostatic forces are directly related to the objects' net charges, and inversely related to the distance between the objects' centers squared. The precise three dimensional shapes of biological molecules, like proteins and dna, are maintained by electrostatic interactions between charged amino acid residues or nucleotide bases. these attractions are essential for their function.
Electrostatic Attraction Electrostatic attraction is the force of attraction between oppositely charged particles or objects. it is a fundamental concept in the study of ionic bonding, where the attractive force between positively and negatively charged ions holds the ionic compound together. The second big difference between the electrostatic force and gravity is that electrostatic forces come in two types: attractive and repulsive. there are two types of charge, and that means the force can go in one of two directions: toward and away. meanwhile, gravity is only an attractive force. Explore coulomb's law, which describes the electrostatic forces two electric charges exert on each other. learn how the strengths of the electrostatic forces are directly related to the objects' net charges, and inversely related to the distance between the objects' centers squared. The precise three dimensional shapes of biological molecules, like proteins and dna, are maintained by electrostatic interactions between charged amino acid residues or nucleotide bases. these attractions are essential for their function.

Electrostatic Attraction The Effect Of Metal Shielding Layer On Explore coulomb's law, which describes the electrostatic forces two electric charges exert on each other. learn how the strengths of the electrostatic forces are directly related to the objects' net charges, and inversely related to the distance between the objects' centers squared. The precise three dimensional shapes of biological molecules, like proteins and dna, are maintained by electrostatic interactions between charged amino acid residues or nucleotide bases. these attractions are essential for their function.

Electrostatic Attraction The Effect Of Metal Shielding Layer On
Comments are closed.