Electron Atom Interactions
â ªatomic Interactionsâ Interactions involving electrons with other subatomic particles are of interest in fields such as chemistry and nuclear physics. atoms are composed of positive protons within atomic nuclei and the negative electrons without, held together by coulomb force interaction. Atom electrons, nucleus, bonds: once the way atoms are put together is understood, the question of how they interact with each other can be addressed—in particular, how they form bonds to create molecules and macroscopic materials.
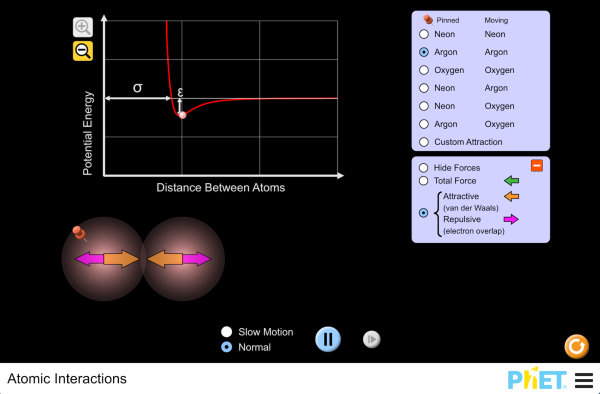
Electron Atom Interactions Atomic interaction refers to the interactions that occur when two atoms approach each other, which are crucial for the formation of chemical bonds. these interactions are influenced by factors such as valence electrons and electronegativity. Our model of the interactions between atoms will involve only electric forces; that is, interactions between electrically charged particles, electrons and protons. in order to understand this we need to recall from physics that when charged particles come close to each other they interact. Explore the interactions between various combinations of two atoms. observe the total force acting on the atoms or the individual attractive and repulsive forces. customize the attraction to see how changing the atomic diameter and interaction strength affects the interaction. The simple model of elastic scattering by coulomb interaction of electrons with the atoms in a material is sufficient to explain basic contrast mechanisms in electron microscopy.

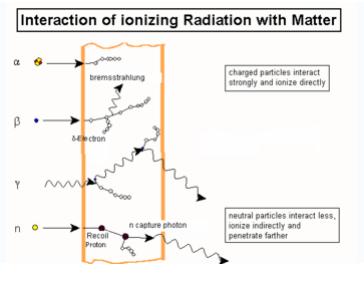
Electron Atom Interactions Explore the interactions between various combinations of two atoms. observe the total force acting on the atoms or the individual attractive and repulsive forces. customize the attraction to see how changing the atomic diameter and interaction strength affects the interaction. The simple model of elastic scattering by coulomb interaction of electrons with the atoms in a material is sufficient to explain basic contrast mechanisms in electron microscopy. In general electrons and positrons lose energy in matter in almost identical ways. there are however some small differences: these contributions are sizeable only for low energies (below about 10 mev) and are never dominant. Atoms interact with one another to achieve a more stable electron configuration, typically by filling their outermost electron shell. this drive for stability results in the formation of chemical bonds, which are lasting attractions between atoms. As the attractions bring the atoms together, electrons from each atom are attracted to the nucleus of both atoms, which “share” the electrons. the sharing of electrons between atoms is called a covalent bond, which holds the atoms together as a molecule. The eii generally, is the interaction of the electrons with a neutral targets (molecules or atoms), which results in formation of a positively charged particles (molecular ions, fragment ions, metastable ions, multiply charged ions ), two or more electrons and also neutral fragments or radicals.

Electron Atom Interactions In general electrons and positrons lose energy in matter in almost identical ways. there are however some small differences: these contributions are sizeable only for low energies (below about 10 mev) and are never dominant. Atoms interact with one another to achieve a more stable electron configuration, typically by filling their outermost electron shell. this drive for stability results in the formation of chemical bonds, which are lasting attractions between atoms. As the attractions bring the atoms together, electrons from each atom are attracted to the nucleus of both atoms, which “share” the electrons. the sharing of electrons between atoms is called a covalent bond, which holds the atoms together as a molecule. The eii generally, is the interaction of the electrons with a neutral targets (molecules or atoms), which results in formation of a positively charged particles (molecular ions, fragment ions, metastable ions, multiply charged ions ), two or more electrons and also neutral fragments or radicals.
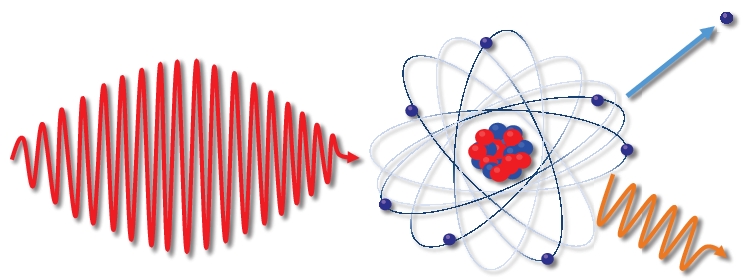
Electron Atom Interactions As the attractions bring the atoms together, electrons from each atom are attracted to the nucleus of both atoms, which “share” the electrons. the sharing of electrons between atoms is called a covalent bond, which holds the atoms together as a molecule. The eii generally, is the interaction of the electrons with a neutral targets (molecules or atoms), which results in formation of a positively charged particles (molecular ions, fragment ions, metastable ions, multiply charged ions ), two or more electrons and also neutral fragments or radicals.
Comments are closed.