Drug Master File Pptx

Drug Master File Pdf Pharmacy Food And Drug Administration A drug master file (dmf) is a confidential submission to the fda containing detailed information about the manufacturing and quality control of human drugs, though it is not mandatory. dmfs support applications like ind, nda, and anda, and help establish the credibility of api manufacturers. It details various types of documents, including master formula records and drug master files, along with their characteristics and preparation procedures. additionally, it discusses the hatch waxman act, which facilitates the approval of generic drugs and provides patent term extensions.

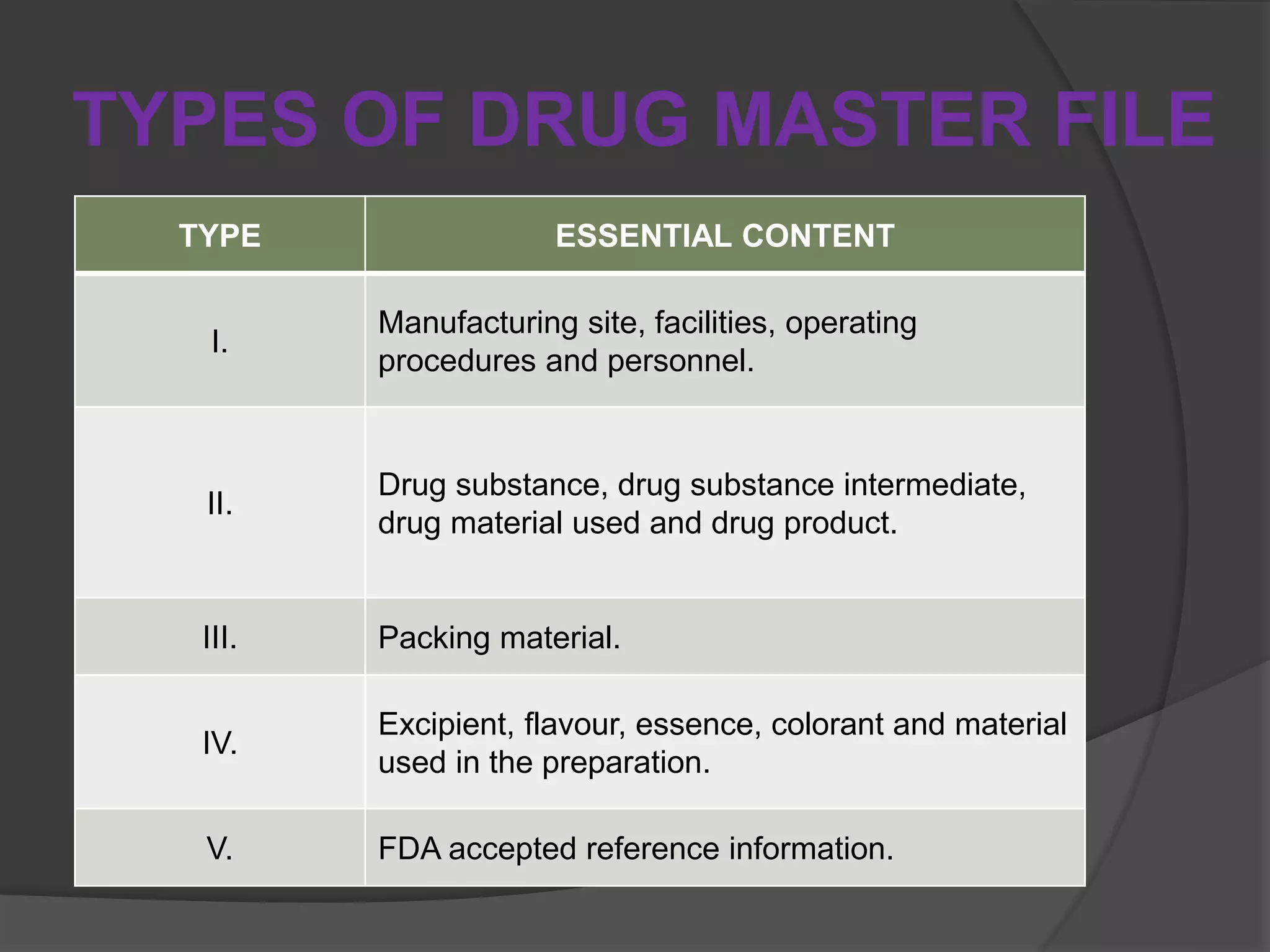
Drug Master File Pdf : drug substance, drug substance intermediate, and material used in their preparation,or drugproduct. 1) drug substance intermediates, drug substances,. The types of dmfs are type ii drug substance, drug substance intermediate, and material used in their preparation; or drug product, type iii packaging material, type iv excipient,. A drug master file (dmf) is a submission to the food and drug administration (fda) that may be used to provide confidential detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of one or more human drugs. It is a document prepared by a manufacturer (holder) to provide the regulatory authority with confidential, detailed information about an active ingredient intended to be used in human or veterinary drugs products.
Drug Master File Pptx A drug master file (dmf) is a submission to the food and drug administration (fda) that may be used to provide confidential detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of one or more human drugs. It is a document prepared by a manufacturer (holder) to provide the regulatory authority with confidential, detailed information about an active ingredient intended to be used in human or veterinary drugs products. The live online training is designed for all persons involved in the compilation of pharmaceutical dossiers for marketing au thorisations especially for drug master files who want to be come familiar with the diferent dmf procedures. A drug master file or dmf is a document prepared by a pharmaceutical manufacturer and submitted solely at its discretion to the appropriate regulatory authority in the intended drug market. The nda application is a formal proposal by drug sponsors to obtain approval for a new pharmaceutical, detailing its clinical tests, ingredients, and manufacturing processes. The document provides a comprehensive overview of drug master files (dmfs) submitted to the fda, detailing types, submission procedures, and obligations of the holder.

Drug Master File Pptx The live online training is designed for all persons involved in the compilation of pharmaceutical dossiers for marketing au thorisations especially for drug master files who want to be come familiar with the diferent dmf procedures. A drug master file or dmf is a document prepared by a pharmaceutical manufacturer and submitted solely at its discretion to the appropriate regulatory authority in the intended drug market. The nda application is a formal proposal by drug sponsors to obtain approval for a new pharmaceutical, detailing its clinical tests, ingredients, and manufacturing processes. The document provides a comprehensive overview of drug master files (dmfs) submitted to the fda, detailing types, submission procedures, and obligations of the holder.

Drug Master File Pptx The nda application is a formal proposal by drug sponsors to obtain approval for a new pharmaceutical, detailing its clinical tests, ingredients, and manufacturing processes. The document provides a comprehensive overview of drug master files (dmfs) submitted to the fda, detailing types, submission procedures, and obligations of the holder.

Drug Master File Pptx
Comments are closed.