Distinguish Between Formic Acid And Acetic Acid The Gravity

Specific Gravity Formic Acid Percent Chart Pdf Alcohol Ethanol Formic acid (hcooh) contains both an aldehydic ( cho) as well as a carboxyl group ( cooh) but acetic acid (ch 3 cooh) contains only a carboxyl group. therefore, formic acid behaves as a reducing agent whereas acetic acid does not. The main difference between formic acid and acetic acid is that formic acid is composed of a carboxyl group attached to a hydrogen atom whereas acetic acid is composed of a methyl group attached to a carboxyl group.

Distinguish Between Formic Acid And Acetic Acid The Gravity In this article, we analyze the differences between formic acid and acetic acid from a professional supplier’s standpoint, helping industry users make informed choices based on their application needs. ### conclusion the key distinguishing reaction is that **formic acid reduces ammonical agno3**, while **acetic acid does not**. this is due to the presence of reducing hydrogen in formic acid. In summary, understanding how to distinguish between formic acid and acetic acid involves examining their chemical structure, physical properties, and chemical reactivity. This article will detail the differences between the two common organic acids, formic acid and acetic acid, including physical and chemical properties, and help you better understand these two acids.

Distinguish Between Formic Acid And Acetic Acid The Gravity In summary, understanding how to distinguish between formic acid and acetic acid involves examining their chemical structure, physical properties, and chemical reactivity. This article will detail the differences between the two common organic acids, formic acid and acetic acid, including physical and chemical properties, and help you better understand these two acids. Understanding how to distinguish formic acid from acetic acid is essential for chemistry students, researchers, and industries that rely on carboxylic acids in production or analysis. in this topic, we’ll explore various ways to differentiate these two acids clearly and effectively. Acetic acid and formic acid are both organic acids commonly found in nature. they have similar chemical structures, with acetic acid having two carbon atoms and formic acid having one carbon atom. however, they differ in their physical properties and uses. Formic acid is generally considered a stronger acid than acetic acid. this is attributed to the electron donating effect of the methyl group in acetic acid, which destabilizes the carboxylate anion formed after deprotonation, making acetic acid less likely to donate a proton compared to formic acid. The difference in boiling point and density between formic acid and acetic acid affects their application in different industrial processes. formic acid has a lower boiling point and a density slightly higher than that of water, which gives it unique advantages in certain extraction processes.
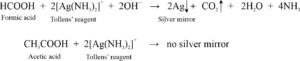
Distinguish Between Formic Acid And Acetic Acid The Gravity Understanding how to distinguish formic acid from acetic acid is essential for chemistry students, researchers, and industries that rely on carboxylic acids in production or analysis. in this topic, we’ll explore various ways to differentiate these two acids clearly and effectively. Acetic acid and formic acid are both organic acids commonly found in nature. they have similar chemical structures, with acetic acid having two carbon atoms and formic acid having one carbon atom. however, they differ in their physical properties and uses. Formic acid is generally considered a stronger acid than acetic acid. this is attributed to the electron donating effect of the methyl group in acetic acid, which destabilizes the carboxylate anion formed after deprotonation, making acetic acid less likely to donate a proton compared to formic acid. The difference in boiling point and density between formic acid and acetic acid affects their application in different industrial processes. formic acid has a lower boiling point and a density slightly higher than that of water, which gives it unique advantages in certain extraction processes.

Distinguish Between Formic Acid And Acetic Acid The Gravity Formic acid is generally considered a stronger acid than acetic acid. this is attributed to the electron donating effect of the methyl group in acetic acid, which destabilizes the carboxylate anion formed after deprotonation, making acetic acid less likely to donate a proton compared to formic acid. The difference in boiling point and density between formic acid and acetic acid affects their application in different industrial processes. formic acid has a lower boiling point and a density slightly higher than that of water, which gives it unique advantages in certain extraction processes.

Distinguish Between Formic Acid And Acetic Acid The Gravity
Comments are closed.