Dissolution Test Apparatus Pptx

Qualification Of Dissolution Test Apparatus Pptx The document provides a comprehensive overview of dissolution testing, its importance in drug formulation, and various dissolution apparatuses used in pharmaceutical research. Dissolutionapparatus free download as powerpoint presentation (.ppt .pptx), pdf file (.pdf), text file (.txt) or view presentation slides online. the document provides an overview of various types of dissolution apparatus used in pharmaceutical testing, including usp, ip, and bp types.
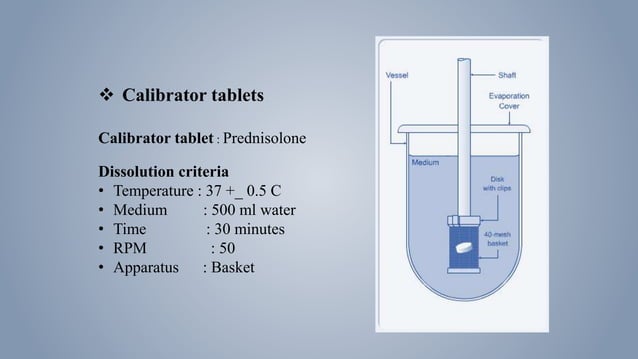
Calibration Of Dissolution Apparatus 1 Pptx The basket is immersed in a dissolution medium (as specified in monograph) contained in a 1000 ml flask. the flask is cylindrical with a hemispherical bottom. the flask is maintained at 37 ± 0.50c by a constant temperature bath. Yatherm scientific: dissolution test apparatus many drug manufacturing companies bring dissolution test apparatus into use to conduct the dissolution testing in manufacturing the bioequivalent oral products. This helps you give your presentation on design and calibration of a dissolution test equipment in a conference, a school lecture, a business proposal, in a webinar and business and professional representations. It outlines the necessary requirements for developing discriminating dissolution methods for fixed dose combination products and emphasizes the need for well established dissolution profiles within physiological ph ranges.

Calibration Of Dissolution Apparatus 1 Pptx This helps you give your presentation on design and calibration of a dissolution test equipment in a conference, a school lecture, a business proposal, in a webinar and business and professional representations. It outlines the necessary requirements for developing discriminating dissolution methods for fixed dose combination products and emphasizes the need for well established dissolution profiles within physiological ph ranges. Dissolution calibration standards are available to make sure that these mechanical and operating requirements are met. rotating paddle (apparatus 2) 13 the paddle apparatus (apparatus ii) consists of a special, coated paddle that minimizes turbulence due to stirring. This document discusses dissolution testing and the various apparatus used. dissolution testing measures how quickly a drug dissolves and is an important standardization process. They are immersed in simulated intestinal fluid for the time specified in the monograph, during which time the tablets disintegrate completely for a positive test. Key factors to consider for dissolution test design are the apparatus, dissolution fluid, and process parameters. the presentation provides details on the assembly, components, and use of each apparatus type.

Dissolution Test Apparatus At 15000 Piece In Ambala Id 2848988687697 Dissolution calibration standards are available to make sure that these mechanical and operating requirements are met. rotating paddle (apparatus 2) 13 the paddle apparatus (apparatus ii) consists of a special, coated paddle that minimizes turbulence due to stirring. This document discusses dissolution testing and the various apparatus used. dissolution testing measures how quickly a drug dissolves and is an important standardization process. They are immersed in simulated intestinal fluid for the time specified in the monograph, during which time the tablets disintegrate completely for a positive test. Key factors to consider for dissolution test design are the apparatus, dissolution fluid, and process parameters. the presentation provides details on the assembly, components, and use of each apparatus type.

Dissolution Test Apparatus At 69000 Pokharan Thane Id They are immersed in simulated intestinal fluid for the time specified in the monograph, during which time the tablets disintegrate completely for a positive test. Key factors to consider for dissolution test design are the apparatus, dissolution fluid, and process parameters. the presentation provides details on the assembly, components, and use of each apparatus type.
Comments are closed.