Direct Biologics Phase 1 Clinical Trial Seeks To Treat Crohn Disease
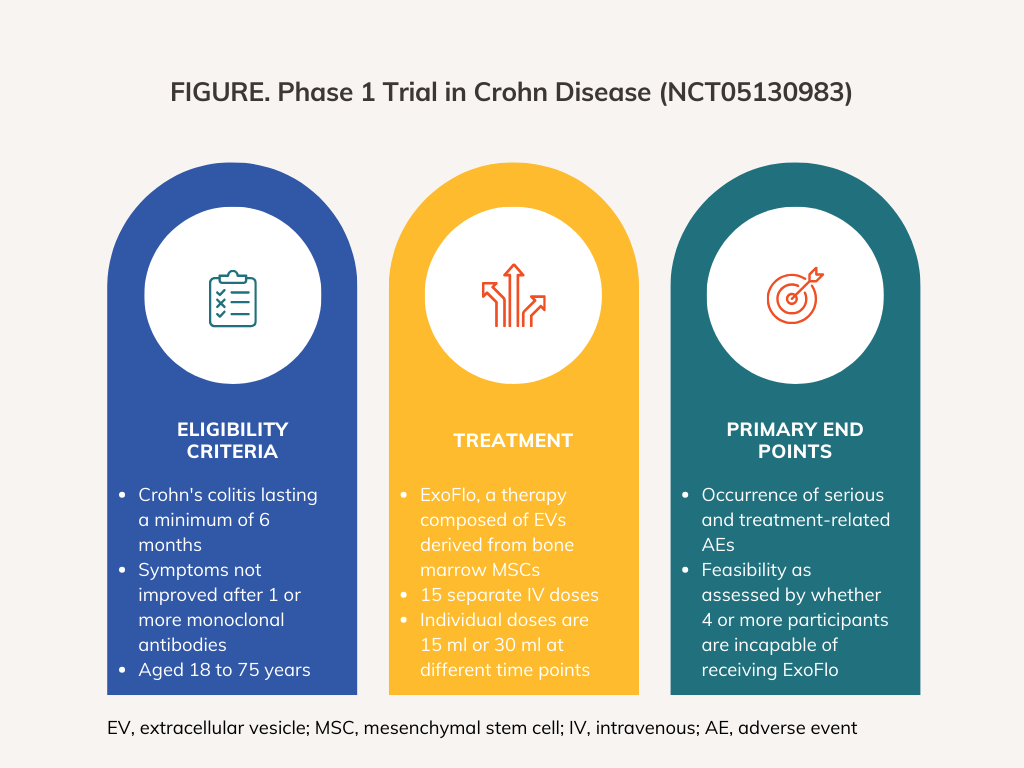
Direct Biologics Phase 1 Clinical Trial Seeks To Treat Crohn Disease Direct biologics receives fda clearance to initiate phase 1b 2a clinical trial of exoflo™ in patients with medically refractory perianal fistulizing crohn’s disease. To evaluate the efficacy of intravenous exoflo in inducing clinical response in subjects with medically refractory crohn's disease who have failed, or are intolerant, or have a contraindication to one or more monoclonal antibodies.

Home Www Directbiologics The open label phase 1 clinical trial (nct05130983) is designed to evaluate the safety and efficacy of exoflo for the treatment of medically refractory crohn’s disease. Title: a phase i study of exoflo, an ex vivo culture expanded adult allogeneic bone marrow mesenchymal stem cell derived extracellular vesicle isolate product, for the treatment of medically refractory crohn's disease. Direct biologics receives fda clearance to initiate phase 1b 2a clinical trial of exoflo™ in patients with medically refractory perianal fistulizing crohn’s disease. The us food and drug administration (fda) has granted clearance to direct biologics’ investigational new drug application to initiate a phase ib iia clinical trial of exoflo to treat medically refractory perianal fistulising crohn’s disease.

Direct Biologics Phase 1 Clinical Trial Seeks To Treat Crohn Disease Direct biologics receives fda clearance to initiate phase 1b 2a clinical trial of exoflo™ in patients with medically refractory perianal fistulizing crohn’s disease. The us food and drug administration (fda) has granted clearance to direct biologics’ investigational new drug application to initiate a phase ib iia clinical trial of exoflo to treat medically refractory perianal fistulising crohn’s disease. Direct biologics given fda clearance to initiate a clinical trial in patients with medically refractory perianal fistulizing crohn’s disease. Direct biologics announces initiation of phase 1 clinical trial for medically refractory crohn’s disease.
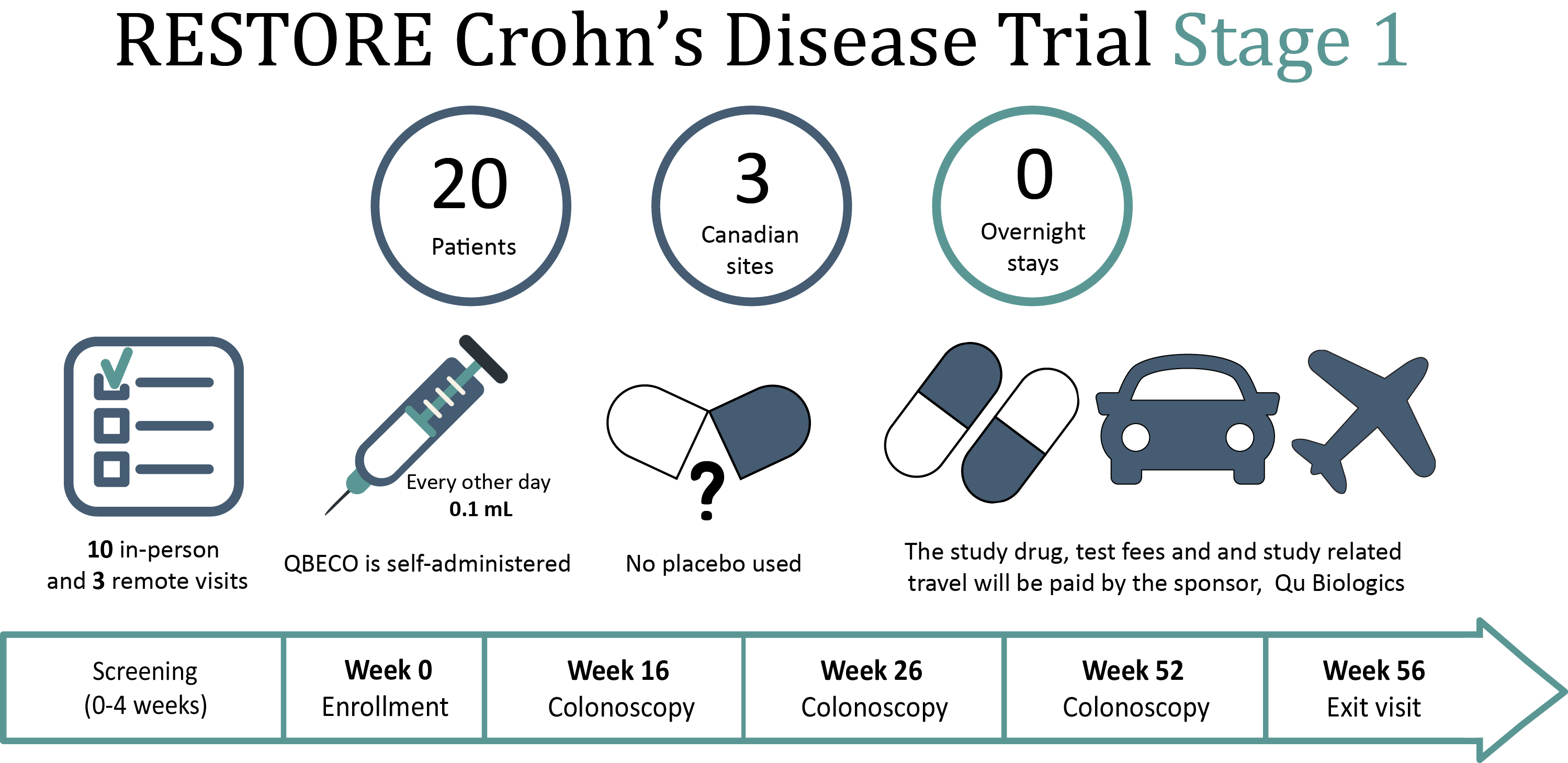
Part 1 Crohn S Disease Phase 2 Clinical Trial Quibd Direct biologics given fda clearance to initiate a clinical trial in patients with medically refractory perianal fistulizing crohn’s disease. Direct biologics announces initiation of phase 1 clinical trial for medically refractory crohn’s disease.

Finding The Right Biologic To Treat Your Crohn S Disease Altus Biologics

Finding The Right Biologic To Treat Your Crohn S Disease Altus Biologics
Comments are closed.