Dilute Vs Concentrated
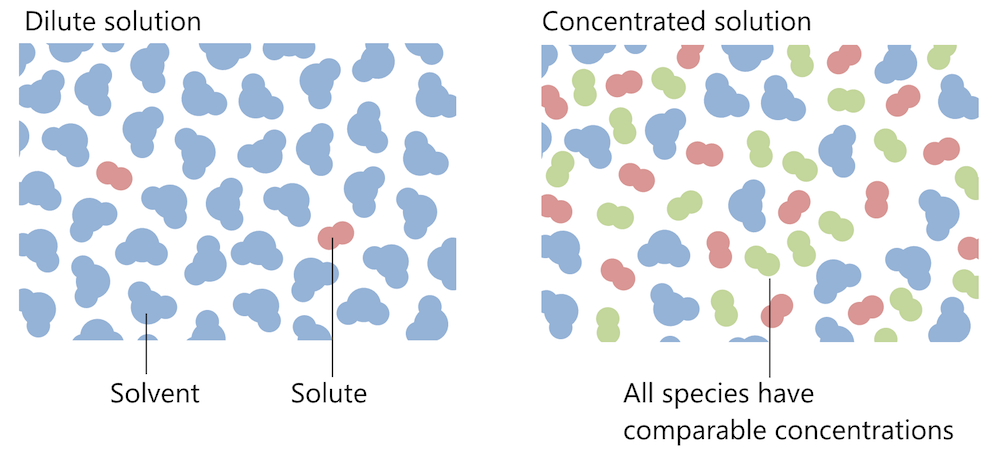
Concentrated Vs Dilute Solutions Learn the difference between dilute and concentrated solutions, two types of homogeneous mixtures. a dilute solution has less solute than a concentrated solution, which has more solute. Learn how to compare and contrast concentrated and dilute substances based on their definition, strength, intensity, properties, uses, and more. find out when to use concentrated or dilute solutions and how they affect chemical reactions and the environment.
Concentrated Vs Dilute Solutions A concentrated solution is one that has a relatively large amount of dissolved solute. a dilute solution is one that has a relatively small amount of dissolved solute. however, these terms are relative, and we need to be able to express concentration in a more exact, quantitative manner. Take care to use the word 'dilute' correctly. it can be used as an adjective to describe the concentration of a solution (as here), or as a verb to describe the process of adding more water to. A strong cup of coffee is a good example of a concentrated solution, while a weak cup of tea represents a dilute solution. similarly, concentrated orange juice needs dilution with water to become a palatable drink, exemplifying the creation of a dilute solution from a concentrated one. Learn the difference between dilute and concentrated solutions in chemistry, with examples, comparison chart, and common curiosities. find out how to measure, alter, and use these solutions in various applications.

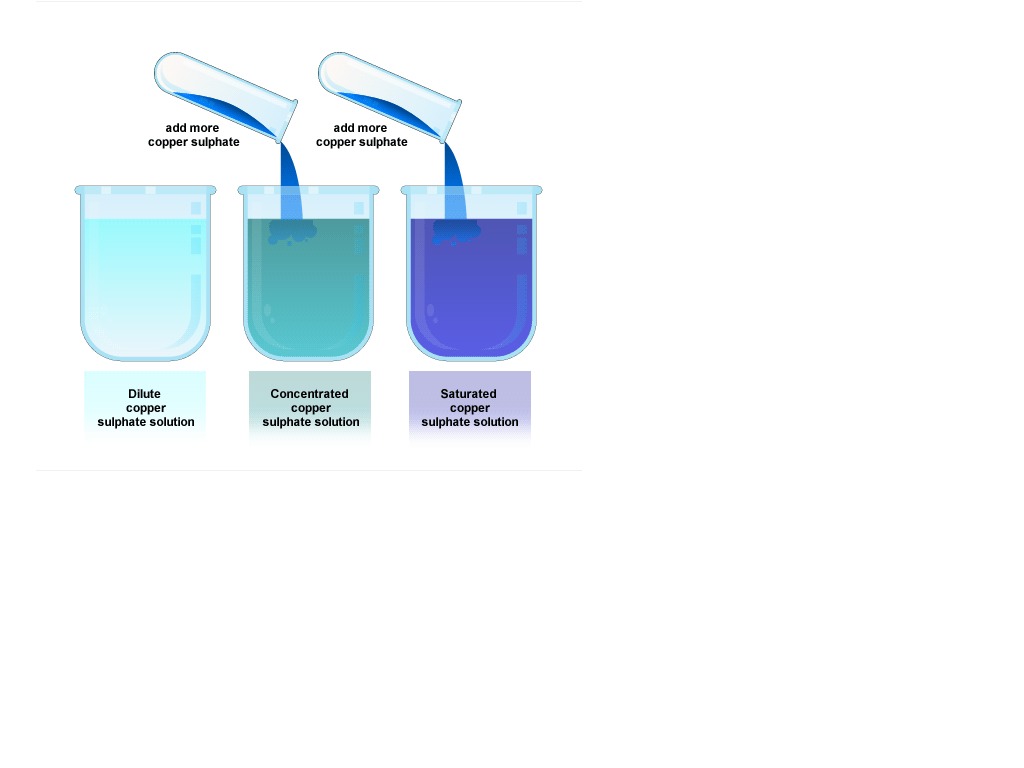
Dilute Solution Vs Concentrated Solution What S The Difference A strong cup of coffee is a good example of a concentrated solution, while a weak cup of tea represents a dilute solution. similarly, concentrated orange juice needs dilution with water to become a palatable drink, exemplifying the creation of a dilute solution from a concentrated one. Learn the difference between dilute and concentrated solutions in chemistry, with examples, comparison chart, and common curiosities. find out how to measure, alter, and use these solutions in various applications. For example, if you add a teaspoon of salt to a liter of water, the resulting solution is a dilute solution of salt in water. concentrated solution: a concentrated solution is one in which a large amount of solute is dissolved in a small amount of solvent. Dilute: to diminish the strength by admixture; usually refers to a solution a solid dissolved in a liquid (weight volume; w v). What is the difference between dilute and concentrated solution? the difference between dilute and concentrated solution is that the amount of solute compared to solvent in a concentrated solution is more than in a dilute solution. Learn the difference between dilute and concentrated solutions based on solute concentration and solubility. see examples of concentrated solutions in liquids, gases and solids and how they change physically and chemically.

Dilute Vs Concentrated Example 1 Video Chemistry Ck 12 For example, if you add a teaspoon of salt to a liter of water, the resulting solution is a dilute solution of salt in water. concentrated solution: a concentrated solution is one in which a large amount of solute is dissolved in a small amount of solvent. Dilute: to diminish the strength by admixture; usually refers to a solution a solid dissolved in a liquid (weight volume; w v). What is the difference between dilute and concentrated solution? the difference between dilute and concentrated solution is that the amount of solute compared to solvent in a concentrated solution is more than in a dilute solution. Learn the difference between dilute and concentrated solutions based on solute concentration and solubility. see examples of concentrated solutions in liquids, gases and solids and how they change physically and chemically.

Science Chemistry Concentrated Vs Dilute Solutions By Jeannie Teacher What is the difference between dilute and concentrated solution? the difference between dilute and concentrated solution is that the amount of solute compared to solvent in a concentrated solution is more than in a dilute solution. Learn the difference between dilute and concentrated solutions based on solute concentration and solubility. see examples of concentrated solutions in liquids, gases and solids and how they change physically and chemically.

Concentrated Vs Dilute Solutions Phet Activity Pdf Concentration
Comments are closed.