Difference Between Oxidation And Reduction Ox Science

Difference Between Oxidation And Reduction Ox Science Oxidation is a reaction where an atom, ion, or molecule loses electrons while reduction corresponds to an atom, ion or molecule gaining electrons. both oxidation and reduction depend on the change in the oxidation state of the atom, that is, on the difference in the charge of the atom in a reaction. Reduction and oxidation occur simultaneously in a type of chemical reaction called a reduction oxidation or redox reaction. the oxidized species loses electrons, while the reduced species gains electrons. despite the name, oxygen need not be present in an oxidation reaction.
Difference Between Oxidation And Reduction Jacks Of Science Oxidation describes the process in which an atom, ion, or molecule loses one or more electrons. conversely, reduction is the process in which a chemical species gains one or more electrons. Oxidation involves the loss of electrons. reduction involves the gain of electrons. oxidation results from the addition of an oxygen atom. reduction results from the removal of oxygen atoms. Oxidation is the loss of hydrogen. reduction is the gain of hydrogen. notice that these are exactly the opposite of the oxygen definitions (#1). for example, ethanol can be oxidized to ethanal: an oxidizing agent is required to remove the hydrogen from the ethanol. While oxidation involves the loss of electrons and an increase in oxidation state, reduction involves the gain of electrons and a decrease in oxidation state. these processes are interconnected and occur simultaneously in redox reactions.
Difference Between Oxidation And Reduction Jacks Of Science Oxidation is the loss of hydrogen. reduction is the gain of hydrogen. notice that these are exactly the opposite of the oxygen definitions (#1). for example, ethanol can be oxidized to ethanal: an oxidizing agent is required to remove the hydrogen from the ethanol. While oxidation involves the loss of electrons and an increase in oxidation state, reduction involves the gain of electrons and a decrease in oxidation state. these processes are interconnected and occur simultaneously in redox reactions. Oxidation is the loss of electrons by a molecule, atom, or ion. reduction is the gain of electrons. these two processes always occur together; when one species is oxidized, another is reduced. one way of remembering which is which is the mnemonic “oil rig”: oxidation is loss, reduction is gain. This lesson introduces oxidation, reduction, and redox reactions for cbse and ncert learning. you identify how substances gain oxygen or lose hydrogen, how reduction removes oxygen or adds hydrogen, and how both processes occur together in redox reactions. What is oxidation and reduction in chemistry? oxidation and reduction are types of chemical reactions where substances exchange electrons. in oxidation, a substance loses electrons (or gains oxygen), while in reduction, a substance gains electrons (or loses oxygen). Oxidation is when an atom loses electrons. reduction is when an atom gains electrons. in oxidation, a molecule or atom loses one or more electrons, and in reduction, a molecule or atom gains one or more electrons.
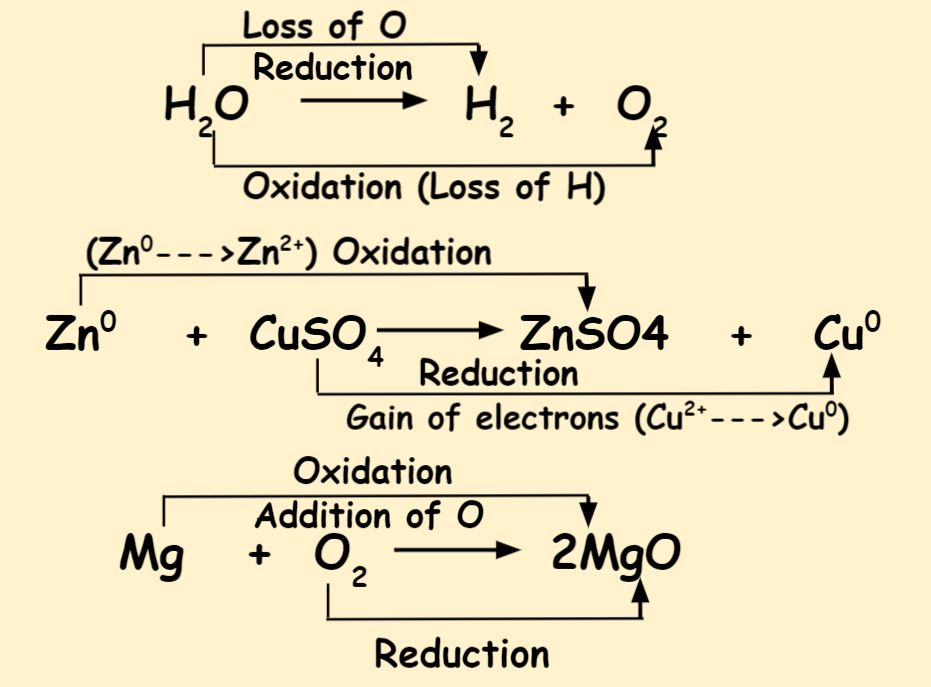
10 Differences Between Oxidation And Reduction Reaction Dewwool Oxidation is the loss of electrons by a molecule, atom, or ion. reduction is the gain of electrons. these two processes always occur together; when one species is oxidized, another is reduced. one way of remembering which is which is the mnemonic “oil rig”: oxidation is loss, reduction is gain. This lesson introduces oxidation, reduction, and redox reactions for cbse and ncert learning. you identify how substances gain oxygen or lose hydrogen, how reduction removes oxygen or adds hydrogen, and how both processes occur together in redox reactions. What is oxidation and reduction in chemistry? oxidation and reduction are types of chemical reactions where substances exchange electrons. in oxidation, a substance loses electrons (or gains oxygen), while in reduction, a substance gains electrons (or loses oxygen). Oxidation is when an atom loses electrons. reduction is when an atom gains electrons. in oxidation, a molecule or atom loses one or more electrons, and in reduction, a molecule or atom gains one or more electrons.

Oxidation And Reduction Classical And Electronic Concept What is oxidation and reduction in chemistry? oxidation and reduction are types of chemical reactions where substances exchange electrons. in oxidation, a substance loses electrons (or gains oxygen), while in reduction, a substance gains electrons (or loses oxygen). Oxidation is when an atom loses electrons. reduction is when an atom gains electrons. in oxidation, a molecule or atom loses one or more electrons, and in reduction, a molecule or atom gains one or more electrons.
Comments are closed.