Difference Between Acetone And Acetate Compare The Difference Between
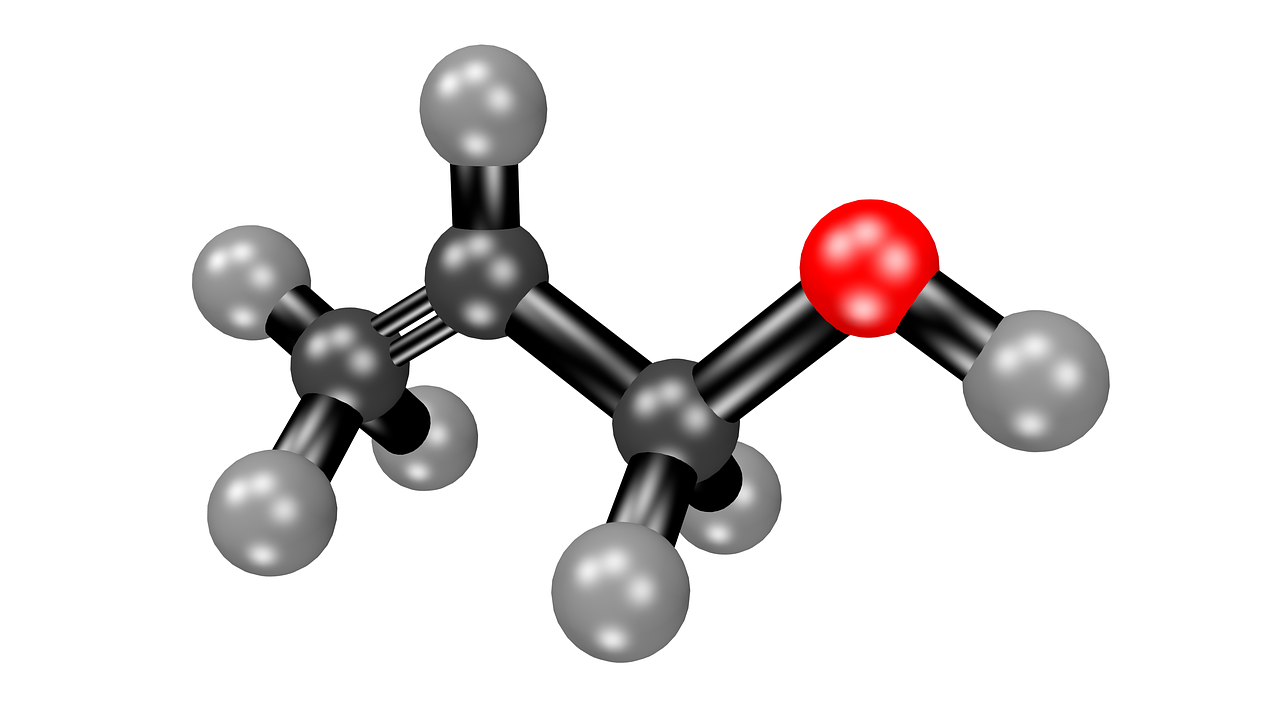
Difference Between Acetone Vs Acetate Difference Between The main difference between acetone and acetate is that acetone is a colorless, volatile organic compound with the chemical formula ch 3 coch 3, whereas acetate is the acetate ion (c 2 h 3 o 2–). Acetate vs. acetone what's the difference? acetate and acetone are both organic compounds that contain the functional group known as the carbonyl group. however, they differ in their chemical structures and properties. acetate, also known as ethanoate, is an anion derived from acetic acid.
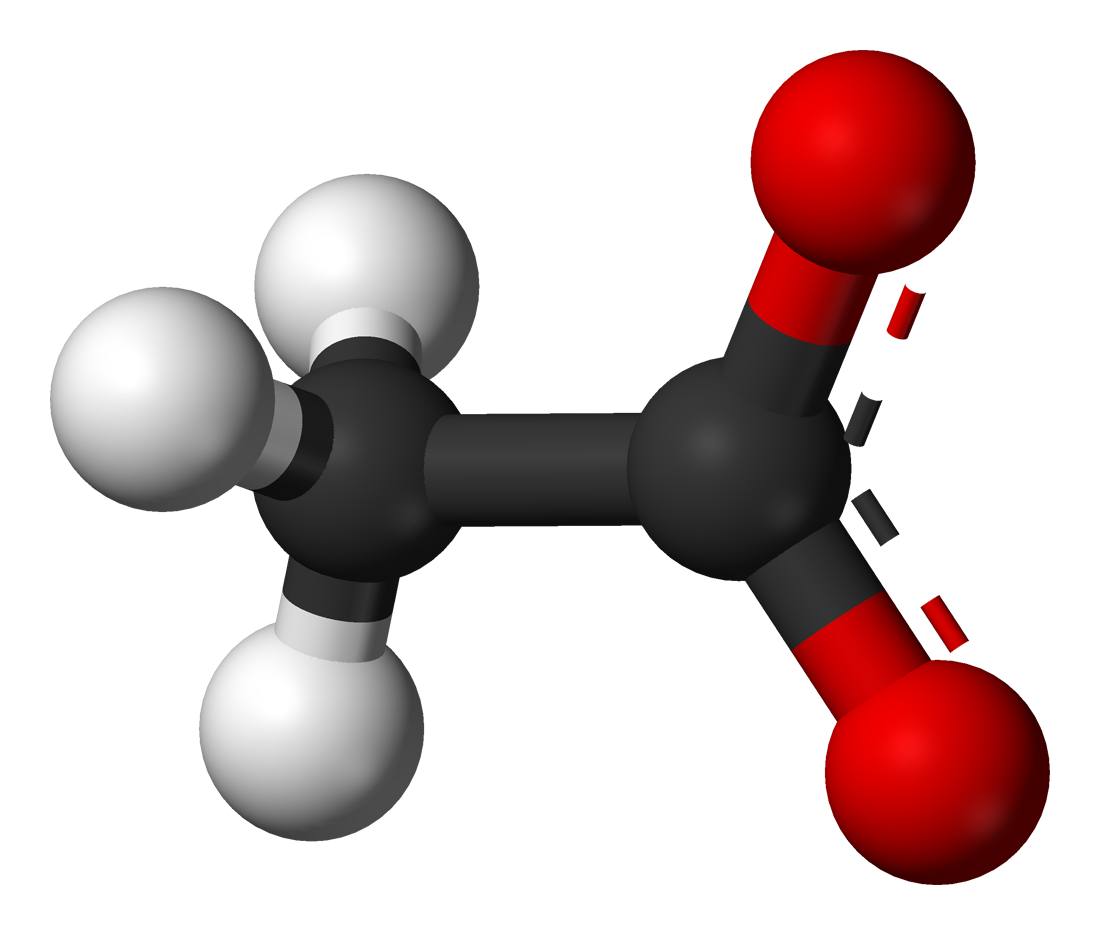
Difference Between Acetone Vs Acetate Difference Between Acetone Despite this phonetic resemblance, acetone and acetate are distinct chemical entities with different structures, properties, and applications. this article aims to clarify the nature of each compound and highlight their fundamental differences. Acetone is an organic compound often used as a solvent; it's the simplest ketone. acetate is an anion derived from acetic acid and is commonly used in making synthetic fibers or plastics. the main difference lies in their chemical structure and uses. In conclusion, acetone and acetate are two distinct compounds that have different chemical structures, properties, and uses. acetone is a volatile and flammable solvent that is commonly used in the manufacture of plastics, fibers, and pharmaceuticals. The primary distinction lies in their molecular composition: acetone is a ketone, containing a carbonyl group (c=o) flanked by two alkyl groups, whereas acetate is a carboxylate, with a negatively charged oxygen atom connected to a carbon atom.

Acetone Vs Acetate Difference And Comparison In conclusion, acetone and acetate are two distinct compounds that have different chemical structures, properties, and uses. acetone is a volatile and flammable solvent that is commonly used in the manufacture of plastics, fibers, and pharmaceuticals. The primary distinction lies in their molecular composition: acetone is a ketone, containing a carbonyl group (c=o) flanked by two alkyl groups, whereas acetate is a carboxylate, with a negatively charged oxygen atom connected to a carbon atom. The key difference between acetone and acetate is that acetone is a naturally occurring compound and acetate is an ingredient created as an additive for other substances. acetate is a common additive in products like cosmetics, cleaning supplies, and textiles. Acetone vs. acetate: what's the difference? acetone is a volatile, flammable solvent, commonly used as a nail polish remover, while acetate is a salt or ester of acetic acid, used in various applications like plastics and textiles. Acetone is a ketone that is commonly used in nail polish remover and even as a paint stripper. acetate is an anion which can also be used to remove paint from surfaces and is commonly combined with molecules to form other useful substances. Acetone is an organic compound while acetate is anion derived from acetic acid (a carboxylic acid). the key difference between acetone and acetate is that acetone is ketone whereas acetate is an anion derived from acetic acid.
Comments are closed.