Data Requirements For Biosimilars
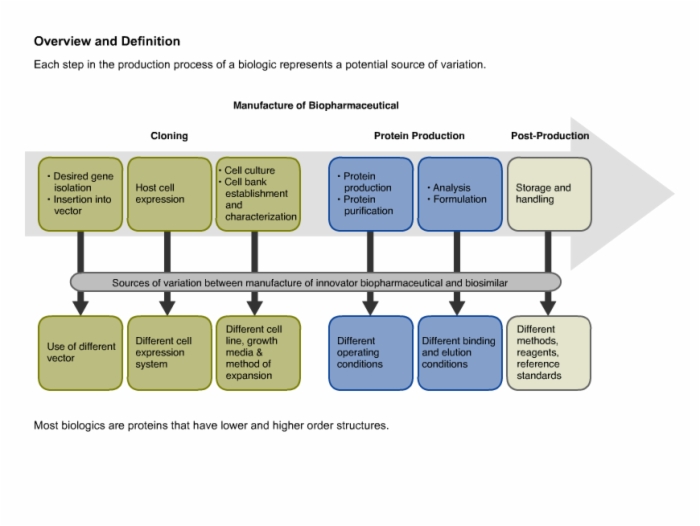
Biotech Biosimilars This page lists biosimilars guidance documents. Generally, non clinical data for a biosimilar are generated through an abbreviated testing programme or in vivo animal studies, as required by the ema’s guidelines.

Biotech Biosimilars These who guidelines are intended to provide globally acceptable principles for the licensing of biological products that are claimed to be similar to biological products of assured quality, safety and efficacy that have been licensed based on a full licensing dossier. We analyze the scientific principles underlying biosimilar development, comparative analytical and clinical requirements, post market surveillance obligations, and emerging regulatory trends. Ema has published scientific guidelines to help developers of biosimilar medicines conform to the strict regulatory requirements for approving biosimilars and to provide transparency on the requirements for approving biosimilars. Regulatory approval of biosimilar medicines currently requires a combination of physicochemical and functional testing, pharmacokinetic data, and clinical efficacy studies (ces).
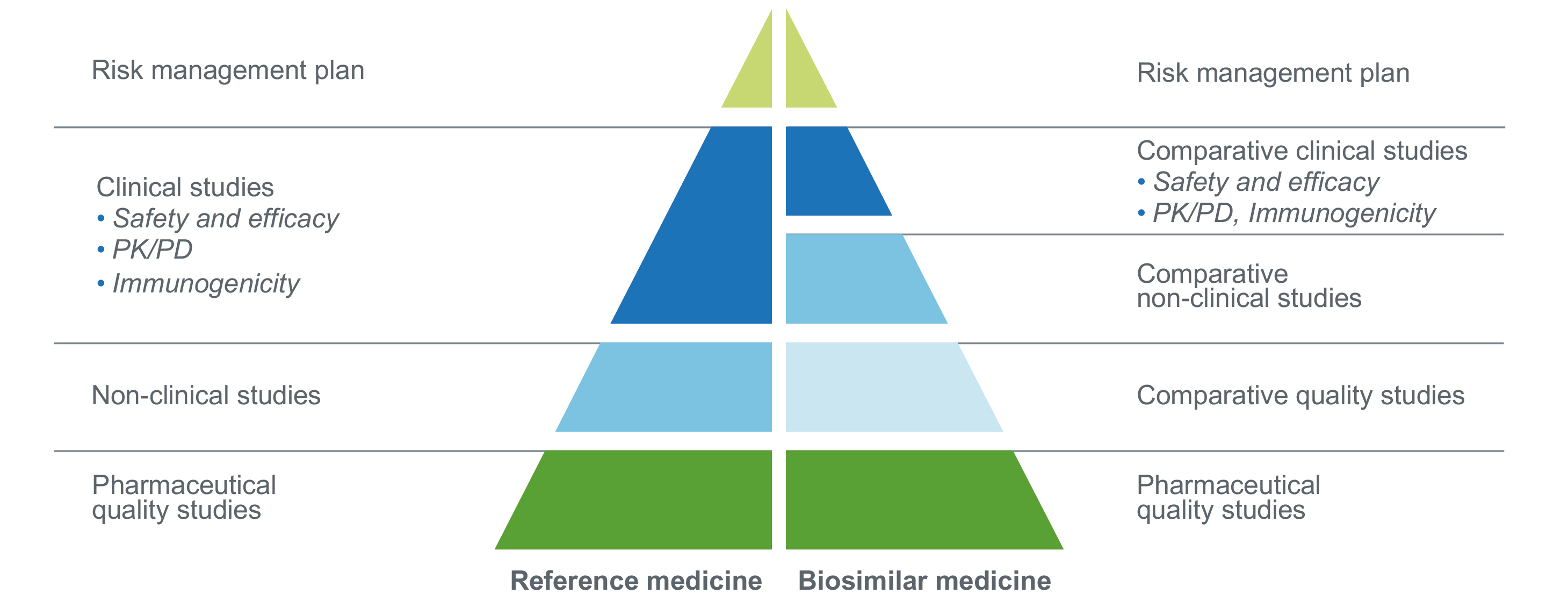
What Are Biosimilars Ema has published scientific guidelines to help developers of biosimilar medicines conform to the strict regulatory requirements for approving biosimilars and to provide transparency on the requirements for approving biosimilars. Regulatory approval of biosimilar medicines currently requires a combination of physicochemical and functional testing, pharmacokinetic data, and clinical efficacy studies (ces). To be approved as a biosimilar, products must demonstrate high similarity to the reference biologic in manufacturing quality, biologic activity, clinical safety and effi cacy, and in the rate of immune reactions. specifi c clinical studies are required to demonstrate this equivalence. Molecule complexity, therapeutic context, and reference product familiarity will dictate data requirements. leveraging cross jurisdiction insights such as oncology or ophthalmology case studies can strengthen submissions. It is the purpose of this article to discuss the numerous regulatory requirements for biosimilar clearance, including the who and several areas, such as india and the eu, as well as the us. Unlike generics, biosimilars are not exact copies due to the biological variability of living systems used in their production. the goal is to match the reference product as closely as possible across a comprehensive range of analytical and clinical parameters.

Relative Data Requirements For Novel Biologics And Biosimilars The To be approved as a biosimilar, products must demonstrate high similarity to the reference biologic in manufacturing quality, biologic activity, clinical safety and effi cacy, and in the rate of immune reactions. specifi c clinical studies are required to demonstrate this equivalence. Molecule complexity, therapeutic context, and reference product familiarity will dictate data requirements. leveraging cross jurisdiction insights such as oncology or ophthalmology case studies can strengthen submissions. It is the purpose of this article to discuss the numerous regulatory requirements for biosimilar clearance, including the who and several areas, such as india and the eu, as well as the us. Unlike generics, biosimilars are not exact copies due to the biological variability of living systems used in their production. the goal is to match the reference product as closely as possible across a comprehensive range of analytical and clinical parameters.
Comments are closed.