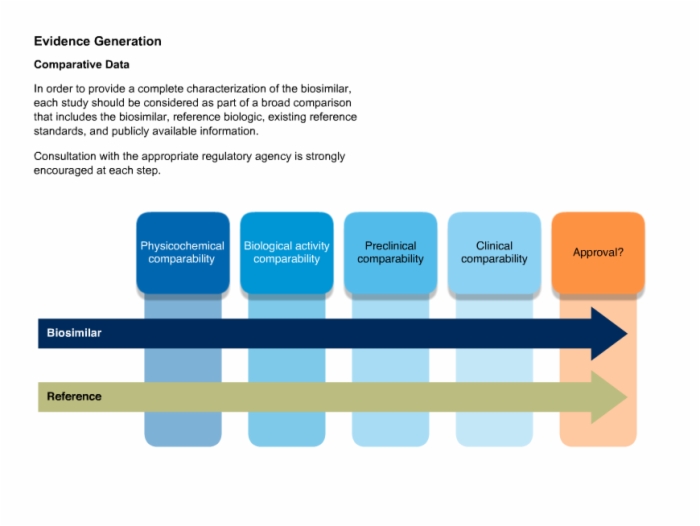
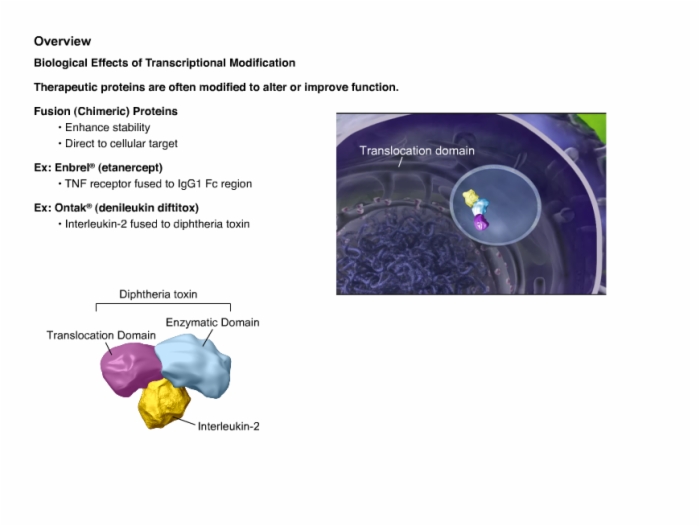
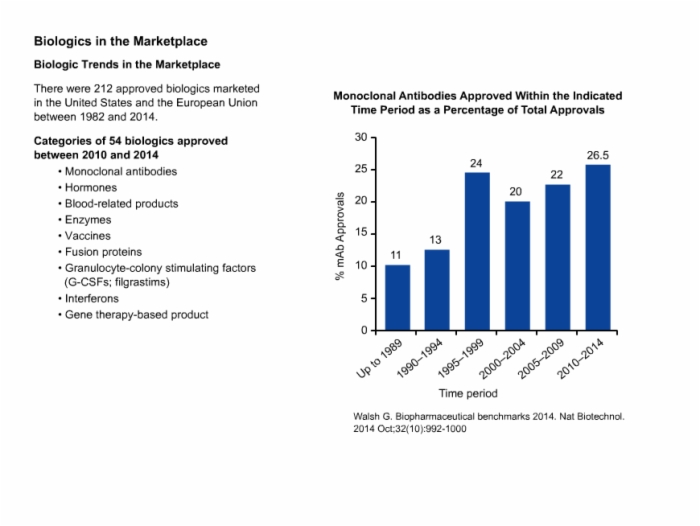
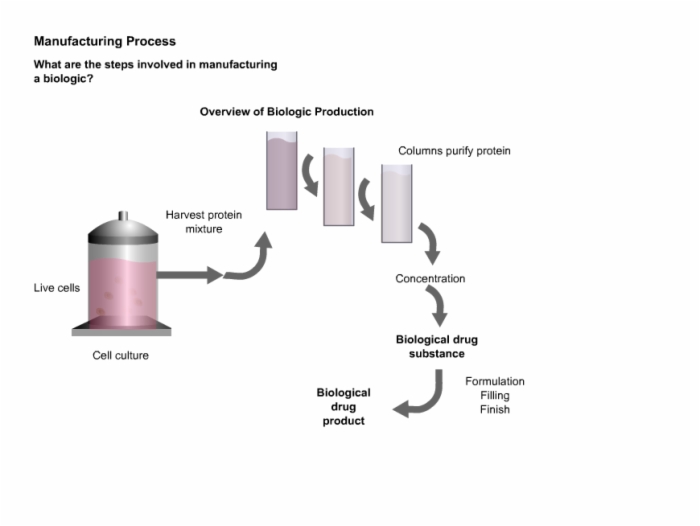
Biotech Biosimilars
Biotech Biosimilars As the patent and exclusivity period for biological medicines draws to a close, there is a possibility for the development and authorization of biosimilars. these products boast comparable levels of safety, quality, and effectiveness to their precursor reference products. Here are 10 leading companies that are shaping the future of the global biosimilars market. headquarters: new york, usa | founded: 1849. pfizer entered the biosimilars market with the strength of its global infrastructure and clinical expertise.
Biotech Biosimilars After the expiry of the patent of approved recombinant drugs (e.g., insulin, human growth hormone, interferons, erythropoietin, monoclonal antibodies and more) any other biotech company can develop and market these biologics (thus called biosimilars). Case studies of biosimilars such as rituximab, adalimumab, filgrastim, and trastuzab reveal both advancements and ongoing hurdles in achieving broader market integration. Biologic medicines have transformed treatment for numerous chronic and life threatening conditions such as cancer, auto immune diseases, and diabetes. these therapies can offer significant benefits in efficacy, safety, and convenience, compared to conventional medicines. The future of biosimilars is bright, with opportunities expanding beyond traditional monoclonal antibodies to include complex proteins, cell therapies, and even gene therapies.
Biotech Biosimilars Biologic medicines have transformed treatment for numerous chronic and life threatening conditions such as cancer, auto immune diseases, and diabetes. these therapies can offer significant benefits in efficacy, safety, and convenience, compared to conventional medicines. The future of biosimilars is bright, with opportunities expanding beyond traditional monoclonal antibodies to include complex proteins, cell therapies, and even gene therapies. Biologics are produced by cells in culture or whole organisms, which are inherently more variable than chemical synthesis methods. therefore, unlike generic pharmaceuticals, it is impossible to. This review addresses the regulatory and manufacturing requirements for biosimilars and provides clinicians with relevant insights for informed prescribing. Daily news and analysis of the global biotechnology, pharmaceutical and medical technology industries. In the meantime, leading innovator biotech companies such as biogen idec and amgen are proposing plans to develop biosimilars, which has mainly called out emerging markets in asia and south america.
Biotech Biosimilars Biologics are produced by cells in culture or whole organisms, which are inherently more variable than chemical synthesis methods. therefore, unlike generic pharmaceuticals, it is impossible to. This review addresses the regulatory and manufacturing requirements for biosimilars and provides clinicians with relevant insights for informed prescribing. Daily news and analysis of the global biotechnology, pharmaceutical and medical technology industries. In the meantime, leading innovator biotech companies such as biogen idec and amgen are proposing plans to develop biosimilars, which has mainly called out emerging markets in asia and south america.
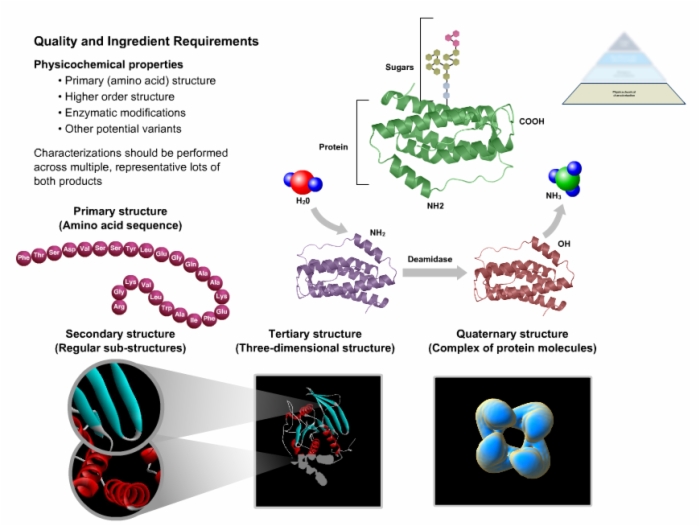
Biotech Biosimilars Daily news and analysis of the global biotechnology, pharmaceutical and medical technology industries. In the meantime, leading innovator biotech companies such as biogen idec and amgen are proposing plans to develop biosimilars, which has mainly called out emerging markets in asia and south america.
Comments are closed.