Crystallization Science Behind Forming Crystals

Crystallization Formation Of Crystals Click Here To Find Out Why Slow The crystallization process begins with nucleation, the initial step where a small number of particles (that is, ions, atoms, or molecules) come together to form a stable cluster of solid material. this cluster acts as a seed for further growth. Crystallization, a fundamental process in nature and industry, is pivotal in forming structured solids from liquids or gases. it involves the orderly arrangement of atoms, ions, or molecules into a highly organized, repeating pattern known as a crystal lattice.

Science Experiment With Crystals Stock Image Image Of Equipment Crystallization occurs extensively within the earth, where minerals form from cooling molten rock, specifically magma or lava. this high temperature process creates igneous rocks, and the resulting crystal size provides a direct record of the cooling conditions. Crystallization is an excellent purification technique for solids because a crystal slowly forming from a saturated solution tends to selectively incorporate particles of the same type into its crystal structure (modeled below). Crystallization occurs in two main phases. the first is nucleation, the appearance of a crystalline phase from either a supercooled liquid or a supersaturated solvent. the second step is known as crystal growth, which is the increase in the size of particles and leads to a crystal state. Initial crystallization creates small nuclei, many of which continue as the centers of crystals during continued growth. because of high temperatures and the molten state of magma, atoms are quite mobile and easily move toward the nuclei and to surfaces of growing crystals.
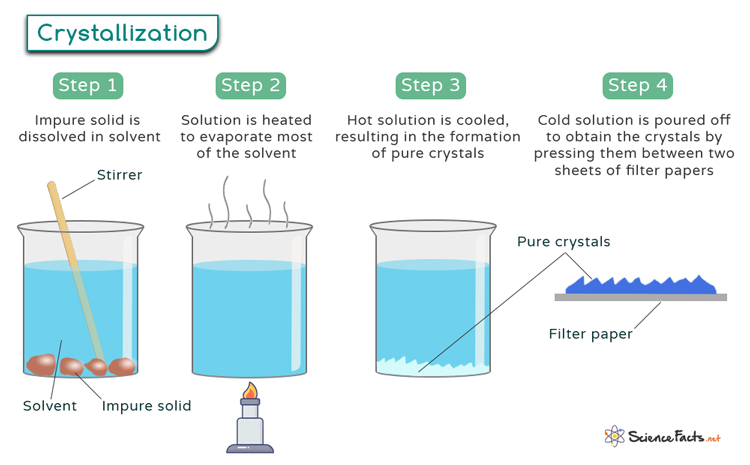
Crystallization Definition Process Examples And Applications Crystallization occurs in two main phases. the first is nucleation, the appearance of a crystalline phase from either a supercooled liquid or a supersaturated solvent. the second step is known as crystal growth, which is the increase in the size of particles and leads to a crystal state. Initial crystallization creates small nuclei, many of which continue as the centers of crystals during continued growth. because of high temperatures and the molten state of magma, atoms are quite mobile and easily move toward the nuclei and to surfaces of growing crystals. The growth of large single crystals is essential to the determination of the structures of molecules and crystals by crystallography and the fabrication of optical and electronic devices. The crystallization process involves two main stages: nucleation, the formation of a seed crystal, and subsequent crystal growth. nucleation can occur spontaneously or be influenced by impurities. Crystallization is the process by which atoms, molecules, or ions arrange themselves into a highly organized, repeating structure called a crystal. it happens when a substance transitions from a disordered state (liquid, gas, or dissolved in solution) into a solid with a fixed geometric pattern. Discover the fascinating process of crystal forming! learn about the types, conditions, and applications of crystals in our comprehensive guide.

Recrystallization Crystals The growth of large single crystals is essential to the determination of the structures of molecules and crystals by crystallography and the fabrication of optical and electronic devices. The crystallization process involves two main stages: nucleation, the formation of a seed crystal, and subsequent crystal growth. nucleation can occur spontaneously or be influenced by impurities. Crystallization is the process by which atoms, molecules, or ions arrange themselves into a highly organized, repeating structure called a crystal. it happens when a substance transitions from a disordered state (liquid, gas, or dissolved in solution) into a solid with a fixed geometric pattern. Discover the fascinating process of crystal forming! learn about the types, conditions, and applications of crystals in our comprehensive guide.
Comments are closed.