Crystallization Process
Crystallization Process Crystallization is the formation of ordered structures from liquids or gases through nucleation, growth, and aggregation. learn how crystallization occurs in nature and industry, and how factors such as temperature, solvent, and impurities influence it. Learn about the process of solidifying atoms or molecules into a crystal lattice, also known as crystallization. explore the different types of crystallization, such as evaporative, cooling, precipitation, and fractional, and their applications in various industries.

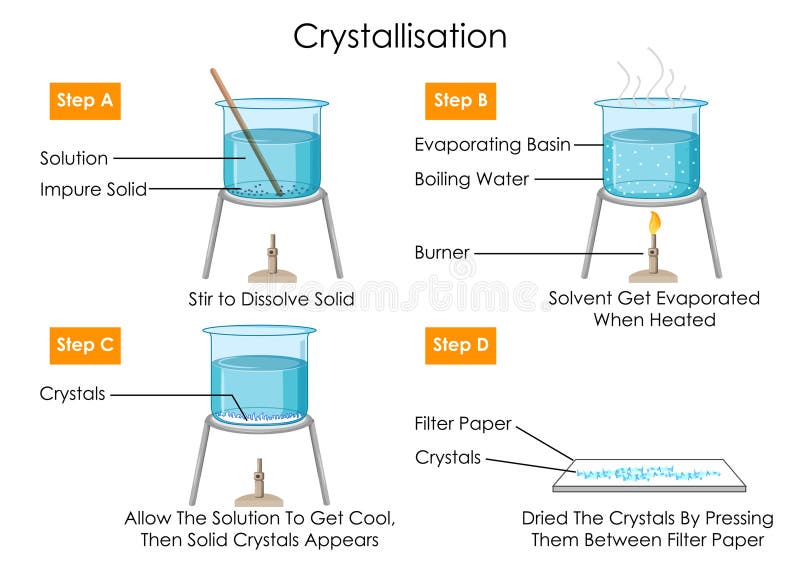
Crystallization Process Crystallization is an excellent purification technique for solids because a crystal slowly forming from a saturated solution tends to selectively incorporate particles of the same type into its crystal structure (modeled below). The crystallization process is defined as a method used for the separation and purification of chemical products, particularly solid products, which allows for the creation of highly pure substances under relatively mild conditions. Crystallization occurs in two main phases. the first is nucleation, the appearance of a crystalline phase from either a supercooled liquid or a supersaturated solvent. the second step is known as crystal growth, which is the increase in the size of particles and leads to a crystal state. Crystallization is a physical process in which a solid forms from a solution, melt, or vapor, creating a highly organized structure called a crystal. this transformation occurs when the solubility conditions change, leading to nucleation and growth of crystal lattices.
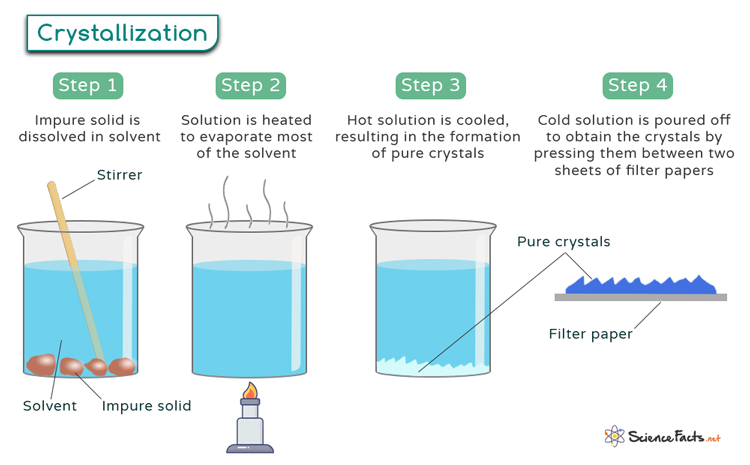
Crystallization Process Industrial Crystallization Process Monitoring Crystallization occurs in two main phases. the first is nucleation, the appearance of a crystalline phase from either a supercooled liquid or a supersaturated solvent. the second step is known as crystal growth, which is the increase in the size of particles and leads to a crystal state. Crystallization is a physical process in which a solid forms from a solution, melt, or vapor, creating a highly organized structure called a crystal. this transformation occurs when the solubility conditions change, leading to nucleation and growth of crystal lattices. Learn about the process of crystallization, where atoms or molecules arrange into a crystal lattice to minimize their energy. find out how to optimize crystal size, yield and purity with different methods and equipment in various industries. Learn about crystallization, its processes, types, and real life uses. clear explanations and examples for students. The crystallization process occurs resulting in a change in the physical property of the liquid, such as the change in temperature, change in its acidity. the crystallization process is carried out on the basis of the size and shapes of the molecules involved, and their chemical properties. Learn what crystallization is, how it works, and why it is important in various fields. explore the process, steps, examples, types, factors, and applications of crystallization with this comprehensive guide.
Comments are closed.