Coordination Chemistry Dpp 03 Of Lec 08 Lakshya Jee 2024 Pdf
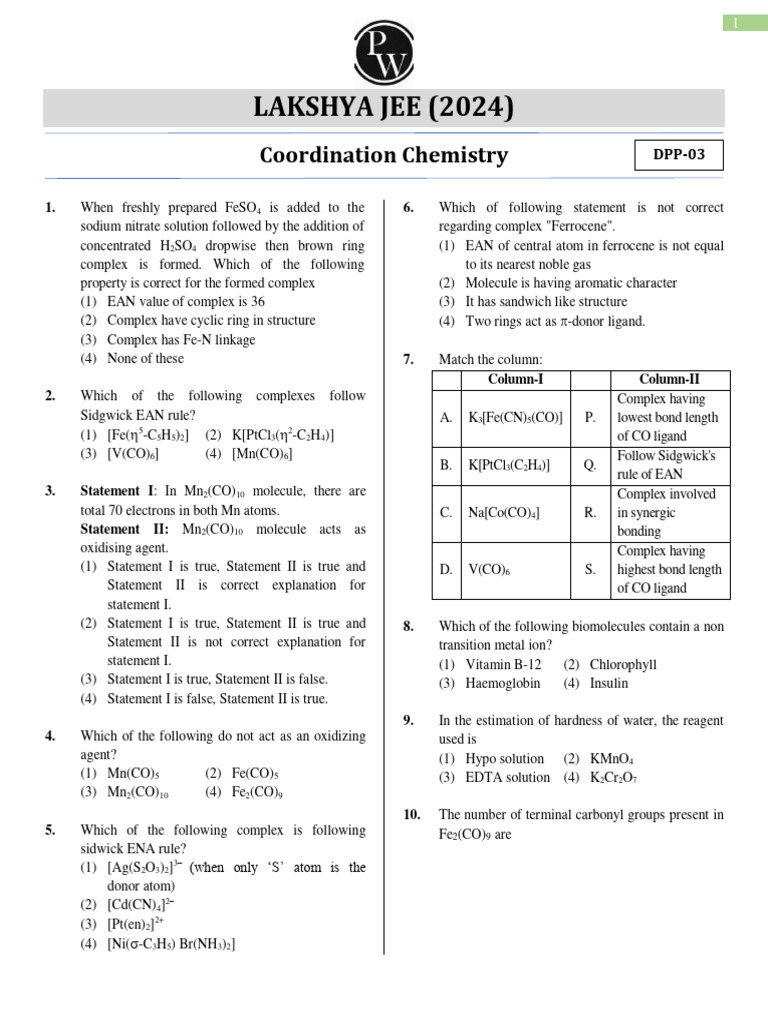
Coordination Chemistry Dpp 03 Of Lec 08 Lakshya Jee 2024 Pdf Coordination chemistry dpp 03 (of lec 08) lakshya jee 2024 free download as pdf file (.pdf), text file (.txt) or read online for free. the document discusses coordination chemistry and provides details about complexes and their properties. Lakshya (jee) coordination compound dpp 03 the coordination number and oxidation state of cr in k3[cr(c2o4)3)] are, respectively 4 and 2 6 and 3.

Lecture Planner Inorganic Chemistry Lakshya Jee 2025 Download Download the study material for jee main exam & jee advanced exam | dlp class | share with your friends google drive. Pdf link . drive.google file d 1qktoejoawxooqurzppeinfhrq5nyic3q view?usp=drivesdkwhole dpp pdf in playlist section. 03 chemical analysis sol chemistry 100% (1) 2 periodic classification dpp 01 (of lec 05) (arjuna jee 2.0 2023) chemistry 100% (1) 2 chemical bonding dpp 01 (of lec 05) (arjuna jee 2.0 2023). Syllabus : coordination compounds 1. which one of the following octahedral complexes will not show geometrical isomerism (a and b are monodentate ligands).

Solution Concentration Calculations Pdf Mass Fraction Chemistry 03 chemical analysis sol chemistry 100% (1) 2 periodic classification dpp 01 (of lec 05) (arjuna jee 2.0 2023) chemistry 100% (1) 2 chemical bonding dpp 01 (of lec 05) (arjuna jee 2.0 2023). Syllabus : coordination compounds 1. which one of the following octahedral complexes will not show geometrical isomerism (a and b are monodentate ligands). We have a huge number of educational video lessons on physics, mathematics, biology & chemistry with concepts & tricks never explained so well before. we upload new video lessons everyday. You can download the coordination compounds notes pdf to study all the topics in this chapter. moreover the class 12 chemistry notes include chapter summary, definitions, examples, and key pointers for coordination compounds. Daily practice problems (chemistry) class xi dpp 01 some basic concepts of chemistry download dpp 02 structure of atom download dpp 03 periodic classification of elements download dpp 04 chemical bonding & molecular structure download dpp 05 states of matter download dpp 06 thermodynamics download dpp 07 equilibrium download dpp 08 redox. Coordination compounds contain complex ions, in which ligands form coordinate bonds to the metal. thus the ligand must have a lone pair of electrons, and the metal must have an empty orbital of suitable energy available for bonding.

Coordination Compounds Dpp 03 Of Lecture 07 Yakeen 3 0 2023 We have a huge number of educational video lessons on physics, mathematics, biology & chemistry with concepts & tricks never explained so well before. we upload new video lessons everyday. You can download the coordination compounds notes pdf to study all the topics in this chapter. moreover the class 12 chemistry notes include chapter summary, definitions, examples, and key pointers for coordination compounds. Daily practice problems (chemistry) class xi dpp 01 some basic concepts of chemistry download dpp 02 structure of atom download dpp 03 periodic classification of elements download dpp 04 chemical bonding & molecular structure download dpp 05 states of matter download dpp 06 thermodynamics download dpp 07 equilibrium download dpp 08 redox. Coordination compounds contain complex ions, in which ligands form coordinate bonds to the metal. thus the ligand must have a lone pair of electrons, and the metal must have an empty orbital of suitable energy available for bonding.

64abad8090cc770018462ada Moving Charges And Magnetism Dpp 02 Daily practice problems (chemistry) class xi dpp 01 some basic concepts of chemistry download dpp 02 structure of atom download dpp 03 periodic classification of elements download dpp 04 chemical bonding & molecular structure download dpp 05 states of matter download dpp 06 thermodynamics download dpp 07 equilibrium download dpp 08 redox. Coordination compounds contain complex ions, in which ligands form coordinate bonds to the metal. thus the ligand must have a lone pair of electrons, and the metal must have an empty orbital of suitable energy available for bonding.

Solutions Dpp 08 Of Lec 09 Lakshya Jee 2024 Pdf Physical
Comments are closed.