Concealer Vs Corrector Tabular Form Electron Affinity Chemical

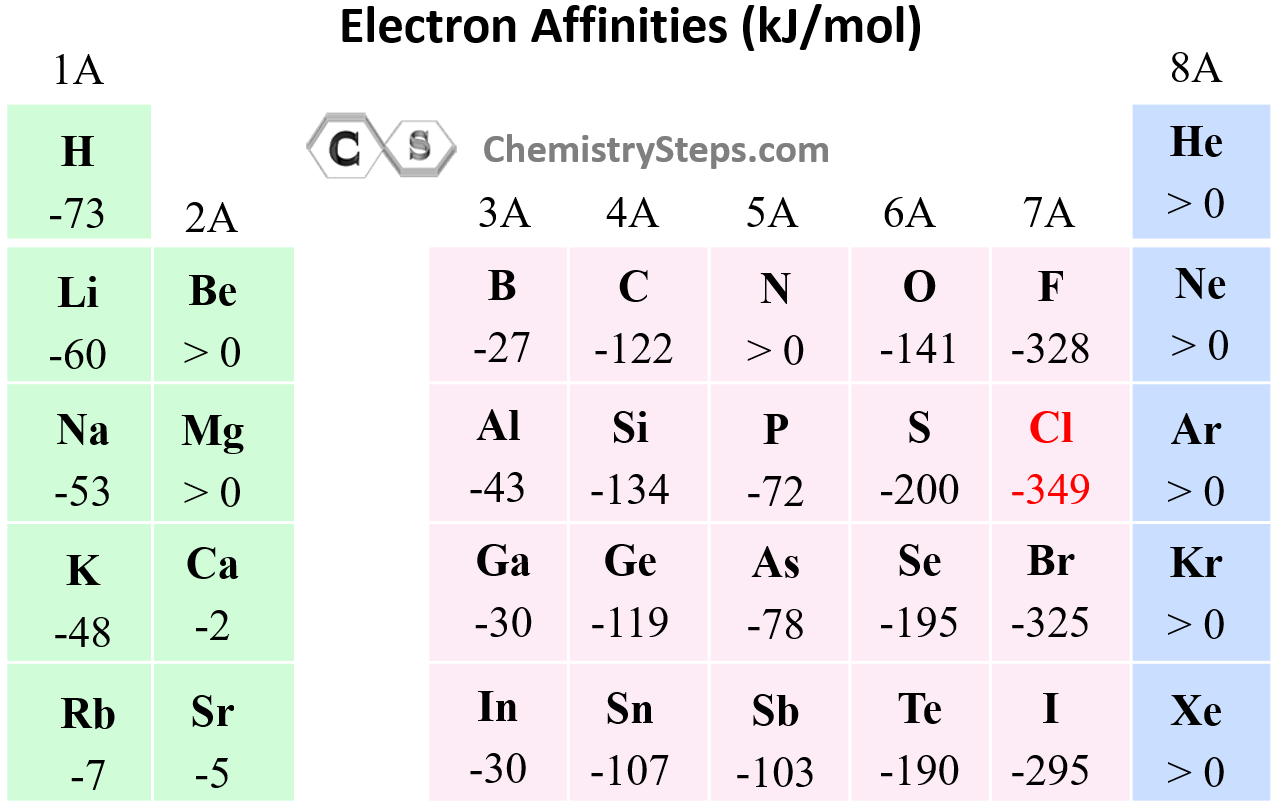
Concealer Vs Corrector Tabular Form Electron Affinity Chemical The relationship of the periodic table with those electron affinities that have been measured or estimated from calculations can be seen on the table of ionization energies and electron affinities (in the red), seen below. Electron affinity chart for all the elements are given below. you can also get the periodic table labeled with electron affinity values of elements.

Electron Affinity Chemistry Steps The electron affinity (eea) of an atom or molecule is defined as the amount of energy released when an electron attaches to a neutral atom or molecule in the gaseous state to form an anion. Learn about electron affinity in chemistry. see the electron affinity trend and learn about the elements with the lowest and highest values. Concealer = cover and blend. corrector = neutralize discoloration. concealers come in skin tone shades to hide blemishes, spots, and under eye shadows. correctors come in color theory hues (green, peach, lavender) to cancel out specific undertones before concealer. for mild imperfections, concealer alone works. The data table shown below lists the electron affinities of the chemical elements as properties of isolated atoms or molecules, i.e. for individual particles in the gas phase.
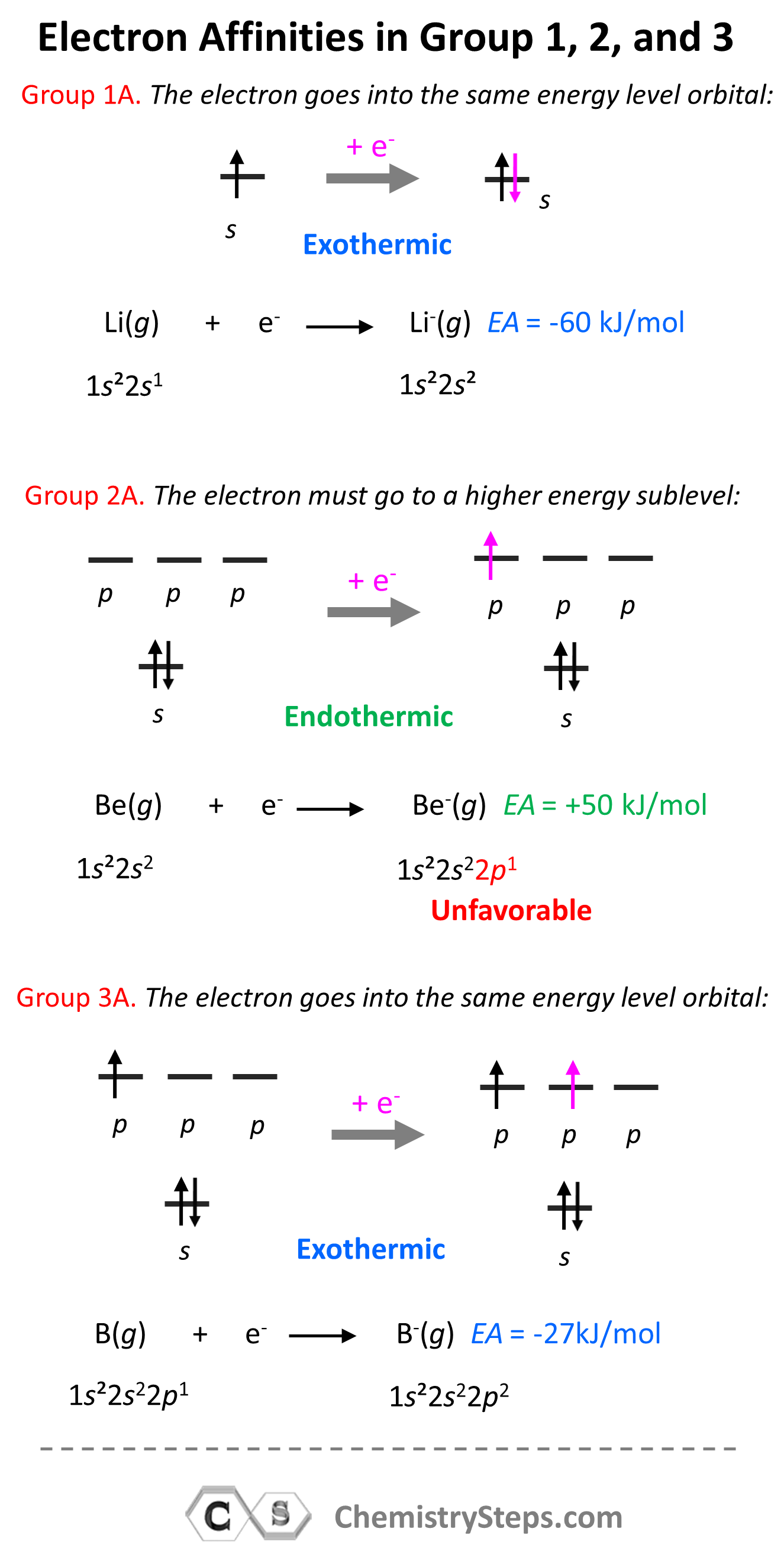
Electron Affinity Chemistry Steps Concealer = cover and blend. corrector = neutralize discoloration. concealers come in skin tone shades to hide blemishes, spots, and under eye shadows. correctors come in color theory hues (green, peach, lavender) to cancel out specific undertones before concealer. for mild imperfections, concealer alone works. The data table shown below lists the electron affinities of the chemical elements as properties of isolated atoms or molecules, i.e. for individual particles in the gas phase. There are two types of electron affinity, first and second. the first involves the addition of an electron to a neutral atom. because this exothermic process releases energy, first electron affinities are negative values. the second pertains to the addition of an electron to a negative ion. Explore how electron affinity changes with atomic number in the periodic table of elements via interactive plots. Electron affinity (the energy associated with forming an anion) is more favorable (exothermic) when electrons are placed into lower energy orbitals, closer to the nucleus. In general, a greater electron affinity implies a lower tendency of the atom to accept an extra electron. in table 9, experimental results of the electron affinities of some neutral clusters are shown, as well as ab initio results.
Electron Affinity Chemistry Steps There are two types of electron affinity, first and second. the first involves the addition of an electron to a neutral atom. because this exothermic process releases energy, first electron affinities are negative values. the second pertains to the addition of an electron to a negative ion. Explore how electron affinity changes with atomic number in the periodic table of elements via interactive plots. Electron affinity (the energy associated with forming an anion) is more favorable (exothermic) when electrons are placed into lower energy orbitals, closer to the nucleus. In general, a greater electron affinity implies a lower tendency of the atom to accept an extra electron. in table 9, experimental results of the electron affinities of some neutral clusters are shown, as well as ab initio results.
Comments are closed.