Compound Toxicity Evaluation Drug Development Process Single
Challenges With Local Toxicity Assessment In Drug Development Toxicological studies are a critical component of pharmaceutical development, and developing effective formulations will help with candidate selection and optimization, and to ensure safety in subsequent human trials. Regulatory toxicology studies are mandatory in the drug development process and aim to evaluate the toxicity level of a substance using protocols that follow the guidelines recommended to conduct non clinical studies of pharmaceutical products.

Compound Toxicity Evaluation Drug Development Process Single This article provides a brief outline of the processes of new drug discovery and development. This review highlights the importance of toxicological evaluation in new chemical entities (nces) as a critical step in drug development. despite reliance on animal models for safety testing, translation to humans remains essential for establishing true safety and efficacy. It typically spans 10–15 years and involves several stages, including drug discovery, preclinical testing, clinical trials, regulatory approval, and post marketing surveillance. Drug discovery typically, researchers discover new drugs by the following methods: through new insights into a disease process that allow researchers to design a product to stop or reverse the effects of the disease.
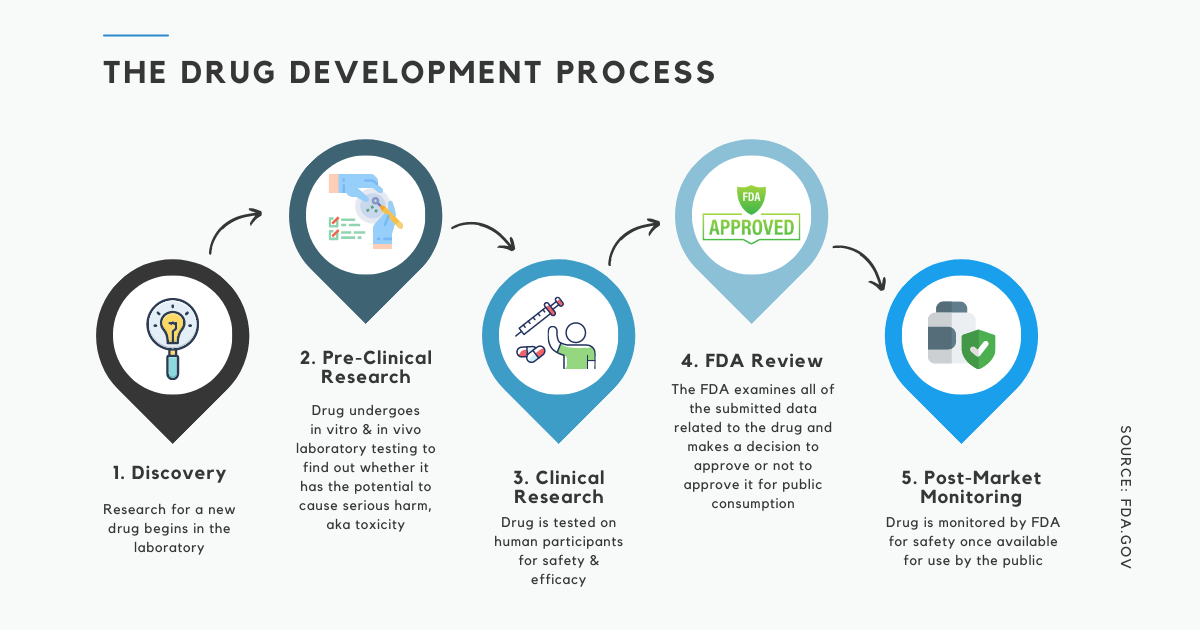
The Importance Of Immunotoxicity Testing In Drug Development Brt Blog It typically spans 10–15 years and involves several stages, including drug discovery, preclinical testing, clinical trials, regulatory approval, and post marketing surveillance. Drug discovery typically, researchers discover new drugs by the following methods: through new insights into a disease process that allow researchers to design a product to stop or reverse the effects of the disease. The primary objective of toxicology studies at preclinical stages is to evaluate the safety of drug candidates before human trials. this chapter provides a comprehensive review of the principles, methodologies, and regulatory requirements that ensure the safety of new therapeutic agents. This article will provide a practical risk based approach for strengthening pharmaceutical safety through toxicological evaluation and associated controls as part of the modern drug development process, particularly within shared facilities. introduction. Learn the complete drug development process, including target identification, lead optimization, preclinical testing, pharmacological and toxicological evaluation, and ind submission. a clear, student friendly guide for pharm.d and clinical research learners. Our experienced team can help you design single dose acute toxicity studies with the right toxicological species, dose level, routes of administration, analyses, and regulatory support – for all stages of the drug development continuum.
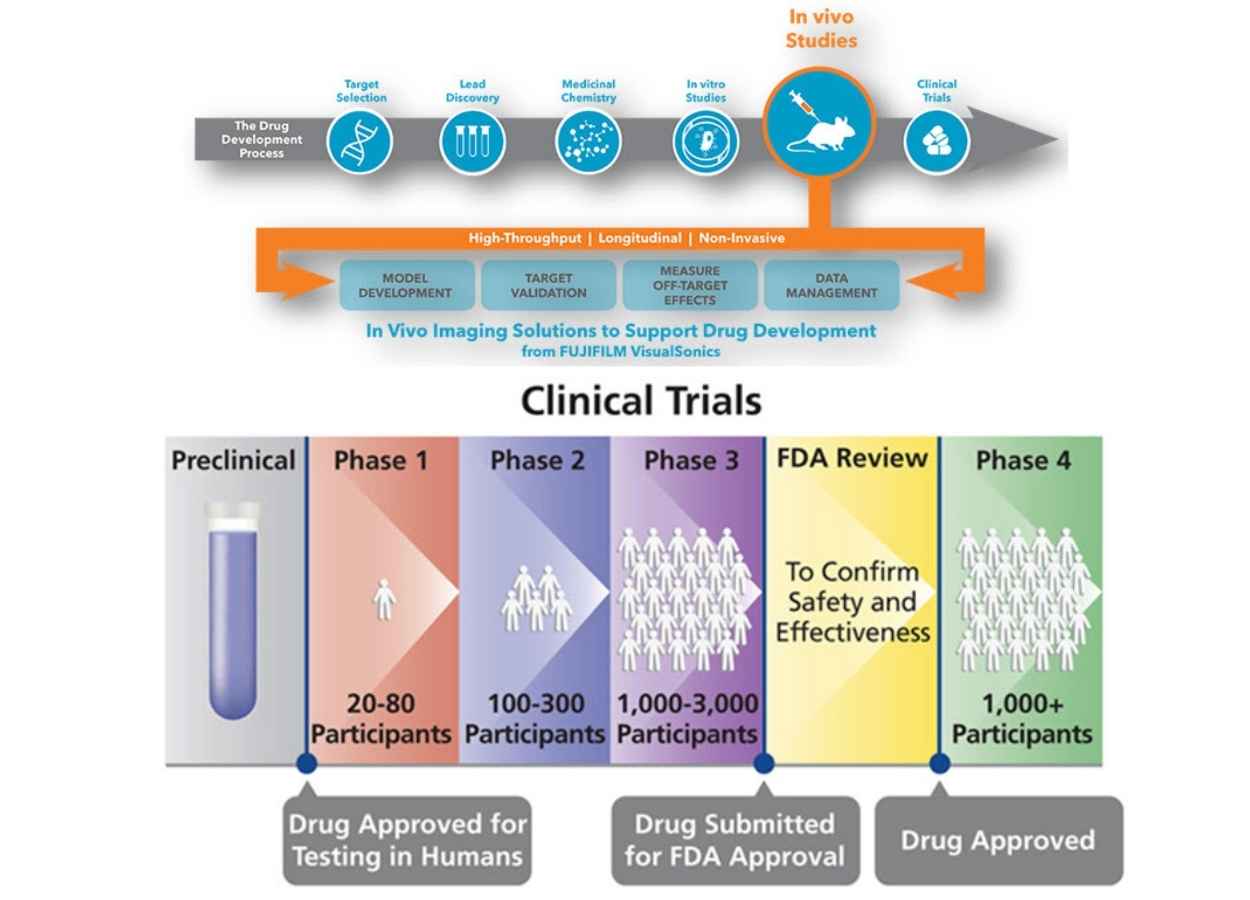
Drug Development Process Overview The primary objective of toxicology studies at preclinical stages is to evaluate the safety of drug candidates before human trials. this chapter provides a comprehensive review of the principles, methodologies, and regulatory requirements that ensure the safety of new therapeutic agents. This article will provide a practical risk based approach for strengthening pharmaceutical safety through toxicological evaluation and associated controls as part of the modern drug development process, particularly within shared facilities. introduction. Learn the complete drug development process, including target identification, lead optimization, preclinical testing, pharmacological and toxicological evaluation, and ind submission. a clear, student friendly guide for pharm.d and clinical research learners. Our experienced team can help you design single dose acute toxicity studies with the right toxicological species, dose level, routes of administration, analyses, and regulatory support – for all stages of the drug development continuum.

Drug Development Process Ppt Learn the complete drug development process, including target identification, lead optimization, preclinical testing, pharmacological and toxicological evaluation, and ind submission. a clear, student friendly guide for pharm.d and clinical research learners. Our experienced team can help you design single dose acute toxicity studies with the right toxicological species, dose level, routes of administration, analyses, and regulatory support – for all stages of the drug development continuum.
Comments are closed.