Combinatorial Chemistry One Step Synthesis Molecular Library Design Using Python Rdkit

Combinatorial Synthesis Edited Pdf This google colab notebook 'combinatorial chemistry (one step synthesis) molecular library design using python & rdkit' is designed to enable you to create your own libraries. Create large virtual libraries of small molecules using real chemistry rules, purchasable building blocks and then analyse the datasets.

Combinatorial Chemistry Pdf Organic Synthesis Chemistry First, we have to run the reaction. let's show it working on isopropanol. a couple of things to note about the input and output to the runreactants() method: the input is a python tuple. note the extra parentheses and the trailing comma in the input above. Clearly here, a grignard reaction makes sense, while a diels alder doesn't. now, how can a model learn this process? in this notebook, we will learn how to extract a library of transformations. Build new molecules from brics fragments. fragments ( ) – a sequence of brics fragments to use for building new molecules. onlycompletemols ( ) – if true, only molecules without attachment points will be yielded. seeds ( ) – an optional list of seed molecules to use as starting points. Beginning with the 2019.03 release, the rdkit is no longer supporting python 2. if you need to continue using python 2, please stick with a release from the 2018.09 release cycle. what is this? ¶ this document is intended to provide an overview of how one can use the rdkit functionality from python. it’s not comprehensive and it’s not a.

Combinatorial And Parallel Synthesis Tb Pdf Build new molecules from brics fragments. fragments ( ) – a sequence of brics fragments to use for building new molecules. onlycompletemols ( ) – if true, only molecules without attachment points will be yielded. seeds ( ) – an optional list of seed molecules to use as starting points. Beginning with the 2019.03 release, the rdkit is no longer supporting python 2. if you need to continue using python 2, please stick with a release from the 2018.09 release cycle. what is this? ¶ this document is intended to provide an overview of how one can use the rdkit functionality from python. it’s not comprehensive and it’s not a. The rdkit is a collection of cheminformatics and machine learning software written in c and python. if you are working in python and using conda (our recommendation), installation is super easy: you can then take a look at our getting started in python guide. more detailed installation instructions are available in docs book install.md. This document provides example recipes of how to carry out particular tasks using the rdkit functionality from python. the contents have been contributed by the rdkit community, tested with the latest rdkit release, and then compiled into this document. This is the approach taken in the rdkit. instead of using patterns to match known aromatic systems, the aromaticity perception code in the rdkit uses a set of rules. the rules are relatively straightforward. aromaticity is a property of atoms and bonds in rings. Molli: a general purpose python toolkit for combinatorial small molecule library generation, manipulation, and feature extraction. the construction, management, and analysis of large in silico molecular libraries is critical in many areas of modern chemistry.

Pdf Combinatorial Chemistry Library Design The rdkit is a collection of cheminformatics and machine learning software written in c and python. if you are working in python and using conda (our recommendation), installation is super easy: you can then take a look at our getting started in python guide. more detailed installation instructions are available in docs book install.md. This document provides example recipes of how to carry out particular tasks using the rdkit functionality from python. the contents have been contributed by the rdkit community, tested with the latest rdkit release, and then compiled into this document. This is the approach taken in the rdkit. instead of using patterns to match known aromatic systems, the aromaticity perception code in the rdkit uses a set of rules. the rules are relatively straightforward. aromaticity is a property of atoms and bonds in rings. Molli: a general purpose python toolkit for combinatorial small molecule library generation, manipulation, and feature extraction. the construction, management, and analysis of large in silico molecular libraries is critical in many areas of modern chemistry.
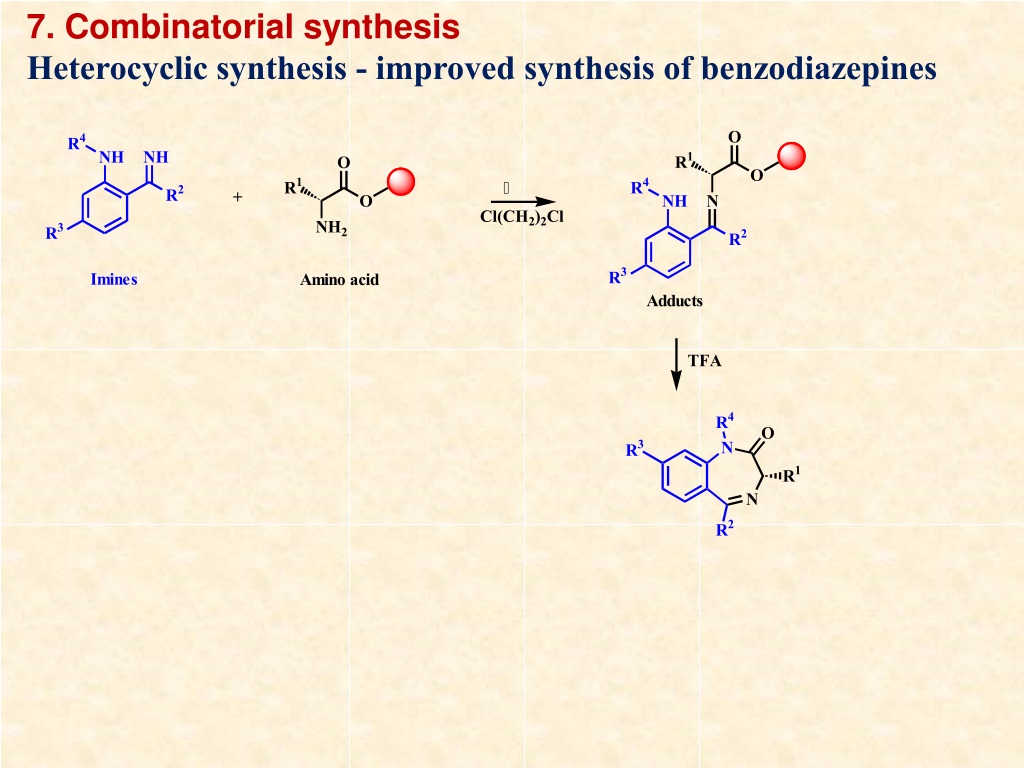
Ppt Combinatorial Chemistry And Library Design By Prof K N Rajini This is the approach taken in the rdkit. instead of using patterns to match known aromatic systems, the aromaticity perception code in the rdkit uses a set of rules. the rules are relatively straightforward. aromaticity is a property of atoms and bonds in rings. Molli: a general purpose python toolkit for combinatorial small molecule library generation, manipulation, and feature extraction. the construction, management, and analysis of large in silico molecular libraries is critical in many areas of modern chemistry.

Ppt Combinatorial Chemistry And Library Design By Prof K N Rajini
Comments are closed.