Clsi Expert Panel Process Changes Overview And Training

Clsi Expert Panel Process Changes Overview And Training Youtube Expert panel volunteers support the development of clsi products by providing technical expertise in specialty areas. learn how you can fill vacancies. Intended for use by clsi expert panels. learn more about recent process changes and training tools.

Basic Steps In A Clinical Expert Panel Cep Download Scientific Diagram From project managers to technical experts, each respective role makes a unique and essential contribution to the creation of clsi’s consensus based standards. it is our goal to sustain and elevate the impact of each valued contribution, while unlocking new efficiencies in standards creation. Approval the revised document is presented to the consensus council, which confirms that the consensus process was followed, and approves the document for publication. Public health and clinical laboratories require a foundation of quality to ensure fidelity in the total testing process. failures at any step can impact patient and population health. find tools to build or enhance your quality management system (qms) on this page. Clsi, the consensus council, and the document development committee on laboratory equipment gratefully acknowledge the expert panel on quality management systems for serving as technical advisors and subject matter experts during the development of this guideline.
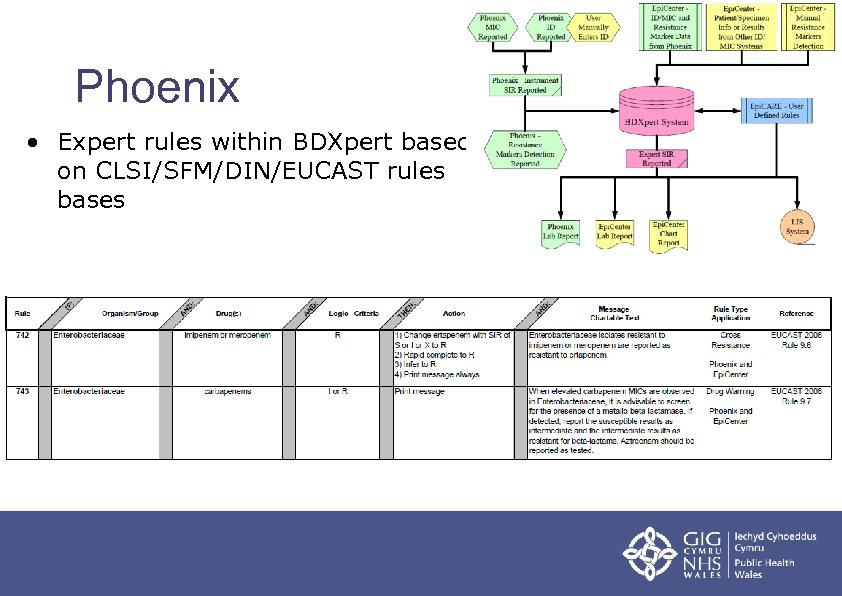
Automated Systems An Overview Robin A Howe Cardiff Public health and clinical laboratories require a foundation of quality to ensure fidelity in the total testing process. failures at any step can impact patient and population health. find tools to build or enhance your quality management system (qms) on this page. Clsi, the consensus council, and the document development committee on laboratory equipment gratefully acknowledge the expert panel on quality management systems for serving as technical advisors and subject matter experts during the development of this guideline. In addition to developing and updating susceptibility breakpoints, the sc develops and validates new testing methods, provides guidance on how results should be interpreted and applied, sets quality control ranges, and educates users through seminars, symposia, and webinars. As of february 12, 2025, unless specific exceptions and additions are identified, fda fully recognizes the standards published in: clinical and laboratory standards institute (clsi) . Standardize the process change through documentation, communication and training. as the new process becomes part of the laboratory quality management system it should be incorporated into the existing continual improvement system. Clsi applied 2026 will provide future ready strategies for clinical and manufacturing lab leaders including ai and technology innovation, standards, and patient safety, all in one powerful half day training event.
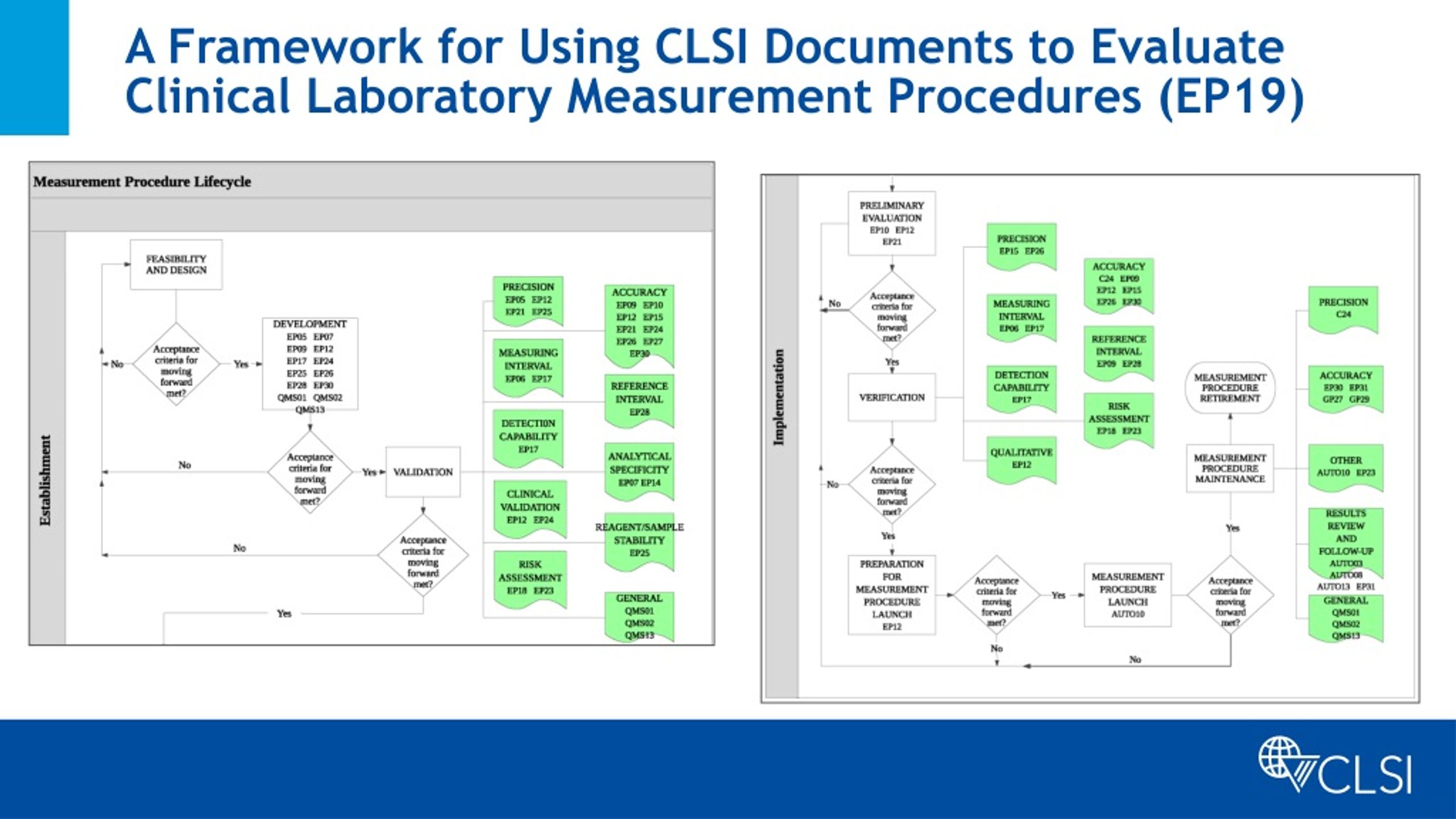
Ppt The Clinical And Laboratory Standards Institute Powerpoint In addition to developing and updating susceptibility breakpoints, the sc develops and validates new testing methods, provides guidance on how results should be interpreted and applied, sets quality control ranges, and educates users through seminars, symposia, and webinars. As of february 12, 2025, unless specific exceptions and additions are identified, fda fully recognizes the standards published in: clinical and laboratory standards institute (clsi) . Standardize the process change through documentation, communication and training. as the new process becomes part of the laboratory quality management system it should be incorporated into the existing continual improvement system. Clsi applied 2026 will provide future ready strategies for clinical and manufacturing lab leaders including ai and technology innovation, standards, and patient safety, all in one powerful half day training event.
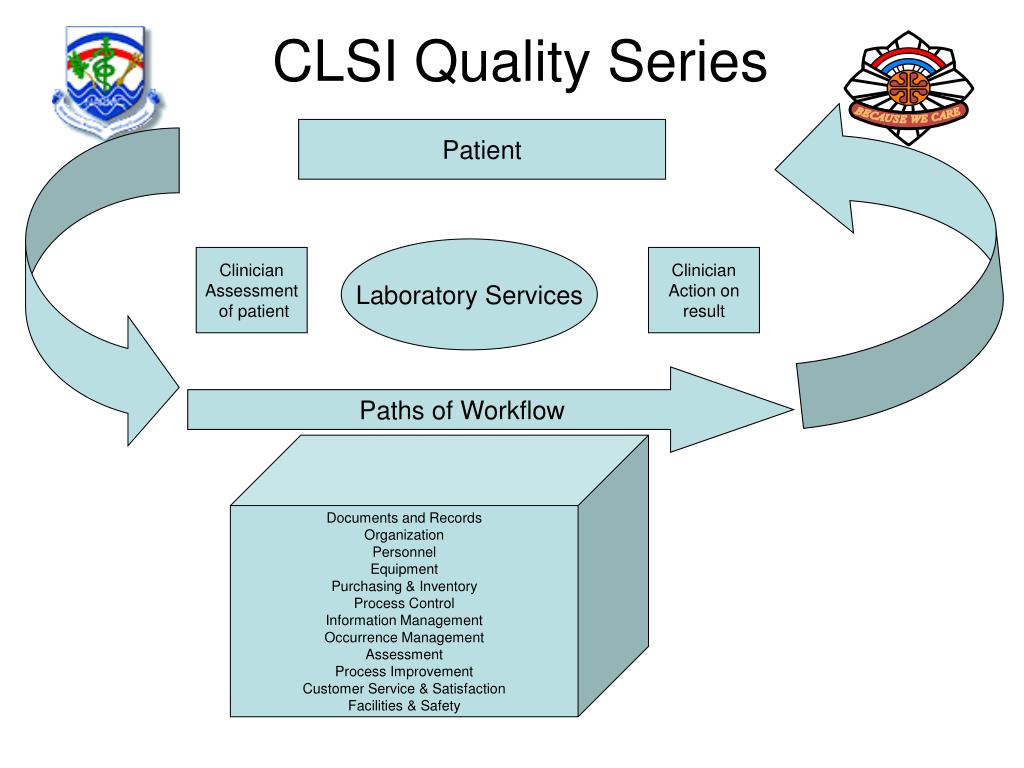
Ppt Ireland Army Community Hospital Powerpoint Presentation Free Standardize the process change through documentation, communication and training. as the new process becomes part of the laboratory quality management system it should be incorporated into the existing continual improvement system. Clsi applied 2026 will provide future ready strategies for clinical and manufacturing lab leaders including ai and technology innovation, standards, and patient safety, all in one powerful half day training event.
Comments are closed.