Clinical Pharmacogenetics Implementation Consortium Guidelines For

Clinical Pharmacogenetics Implementation Consortium Cpic Guidelines Cpic guidelines are designed to help clinicians understand how available genetic test results should be used to optimize drug therapy, rather than whether tests should be ordered. The purpose of this guideline is to provide clinicians with information that facilitates the interpretation of clinical cyp2c19 genotyping test results to guide ppi prescribing.

Clinical Pharmacogenetics Implementation Consortium Cpic Guidelines The guidance emphasises that aminoglycosides should be avoided unless the increased risk of permanent hearing loss is outweighed by the severity of infection and lack of safe or effective alternative therapies. The authors acknowledge the critical input of dr. mary relling and members of the clinical pharmacogenetics implementation consortium (cpic), funded by the national institutes of health. This review of the framework used to assign clinical allele function provides transparency and encourages global participation and feedback from the pharmacogenomics community to promote the adoption of cpic guidelines in clinical practice. This document replaces the 2012 and 2014 clinical pharmacogenetics implementation consortium (cpic) guidelines for slco1b1 and simvastatin induced myopathy.
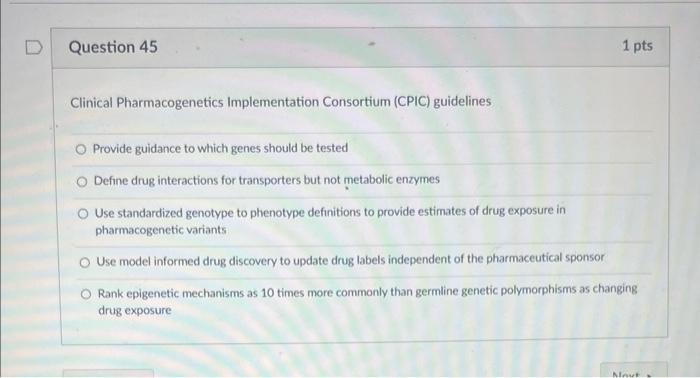
Solved Clinical Pharmacogenetics Implementation Consortium Chegg This review of the framework used to assign clinical allele function provides transparency and encourages global participation and feedback from the pharmacogenomics community to promote the adoption of cpic guidelines in clinical practice. This document replaces the 2012 and 2014 clinical pharmacogenetics implementation consortium (cpic) guidelines for slco1b1 and simvastatin induced myopathy. Understanding cpic guidelines: a guide for clinicians. the clinical pharmacogenetics implementation consortium (cpic) provides clinically relevant, peer reviewed, and up to date guidelines for the use of pharmacogenetic testing in patient care. Cpic's goal is to address this barrier to clinical implementation of pharmacogenetic tests by creating, curating, and posting freely available, peer reviewed, evidence based, updatable, and detailed gene drug clinical practice guidelines (see all cpic publications). The clinical pharmacogenetics implementation consortium (cpic) was established in 2009 as a shared project between pharmgkb and the pharmacogenomics research network (pgrn). pharmgkb and cpic are now clinpgx; cpic continues to publish guidelines and supporting materials as part of clinpgx. read more about cpic. Cpic’s main goal is to provide freely available, evidence based, peer reviewed, and updated pharmacogenetic clinical practice guidelines (1). cpic guidelines have always focused on how to use available pharmacogenetics test results.

Guidelines Of The Clinical Pharmacogenetics Implementation Consortium Understanding cpic guidelines: a guide for clinicians. the clinical pharmacogenetics implementation consortium (cpic) provides clinically relevant, peer reviewed, and up to date guidelines for the use of pharmacogenetic testing in patient care. Cpic's goal is to address this barrier to clinical implementation of pharmacogenetic tests by creating, curating, and posting freely available, peer reviewed, evidence based, updatable, and detailed gene drug clinical practice guidelines (see all cpic publications). The clinical pharmacogenetics implementation consortium (cpic) was established in 2009 as a shared project between pharmgkb and the pharmacogenomics research network (pgrn). pharmgkb and cpic are now clinpgx; cpic continues to publish guidelines and supporting materials as part of clinpgx. read more about cpic. Cpic’s main goal is to provide freely available, evidence based, peer reviewed, and updated pharmacogenetic clinical practice guidelines (1). cpic guidelines have always focused on how to use available pharmacogenetics test results.
Comments are closed.